-
近几十年来,化石燃料燃烧和农业氮肥施用导致区域氮沉降量剧增[1],预计至2050年,全球氮沉积率将达到1995年的2倍[2]。持续的氮输入引发土壤酸化及氮磷比例失衡,影响营养元素的生物地球化学循环,并可能加剧生态系统中的磷限制[3−4]。杉木Cunninghamia lanceolata是中国南方特有的造林和用材树种,因具有较高的经济和生态价值在亚热带林区广泛种植[5]。因此,长期氮沉降背景下杉木人工林的生长可能因土壤氮磷失衡而受到限制。
土壤微生物量碳(MBC)、微生物量氮(MBN)、微生物量磷(MBP)是反映土壤微生物生理状态和养分状况的关键指标。已有研究表明:外源碳氮输入增加使土壤MBP含量增加,表现出土壤微生物对磷的吸收利用[6−7]。然而,现有土壤微生物量研究多局限于碾压破碎、混合均匀的全土尺度,对团聚体微域关注不足。王翠丽等[8]在农田土壤团聚体中发现MBC、MBN含量随团聚体粒径减小而降低;莫雪青[9]对比不同林分土壤团聚体,发现与杉木纯林相比,杉木-红锥Castanopsis hystrix混交林显著提高了各粒径团聚体中的MBN和MBP,而MBC无显著差异。目前,森林土壤团聚体尺度上MBC、MBN、MBP对长期氮添加的响应仍知之甚少。土壤酶是驱动有机质分解和养分循环的生物催化剂,其活性直接影响养分的生物有效性[10]。适量施氮可提高土壤水解酶活性和促进微生物生长[1,11−12],但此类研究同样多以全土为对象,且多关注短期效应。土壤团聚体作为土壤结构的基本单元,形成了物理化学性质各异的微域生境,其通过控制有机质的可接触性、微生物群落结构(如限制微生物与底物的接触)、水分及氧气扩散等因素,对内部的生物、生化过程(包括酶活性)具有隔离和保护作用[13−14]。因此,在长期氮添加背景下,不同粒径团聚体中酶活性及其生态化学计量比的变化特征依然存在不确定性。
本研究基于在福建省三明市沙县官庄国有林场建立的杉木人工林模拟氮沉降长期观测样地,设置不同氮添加水平,聚焦表层土壤不同粒径团聚体,研究长期氮添加下团聚体微生物量碳氮磷、水解酶活性及其生态化学计量特征,并探讨它们之间的相互关系,为评估长期氮沉降对杉木林土壤微域过程的影响提供依据。
-
研究地位于福建省三明市沙县官庄国有林场白溪工区(26°30′N,117°43′E)。该区属亚热带季风气候,全年温暖湿润,光照充足,年均气温为18.8~19.6 ℃,年均降水量为1 606~1 650 mm,无霜期为271 d。试验地土壤为低山丘陵地带发育的山地红壤,平均海拔为200 m,坡向南,坡度为31°。样地所在试验林为1992年由官庄林场统一种植的杉木人工林,总面积为5.173 hm2。林下植被稀疏,主要由五节芒Miseanthus floridulus、芒萁Dicranopteris dichotoma和蕨Pteridium aquilinum等组成,盖度为3%~5%[15]。
-
2003年12月,在立地条件相似的杉木人工林中布设了12块20 m × 20 m的固定标准地。基于亚热带氮沉降显著的地域差异及上升趋势[16],设置了4种氮处理:N0 (0 kg·hm−2·a−1,对照)、N1 (60 kg·hm−2·a−1)、N2 (120 kg·hm−2·a−1)和N3 (240 kg·hm−2·a−1),每处理3个重复标准地。施氮处理始于2004年1月,每月月初使用背负式喷雾器在林地内均匀喷施相应浓度的尿素溶液,对照处理喷施等量水。此施氮处理持续20 a。施氮前进行的林分调查显示:试验林杉木林龄为12 a,林分密度为1 665株·hm−2,平均胸径为16.1 cm,平均树高为12.1 m。2023年6月的林下植被调查显示:N0、N1、N2和N3处理的灌草盖度分别为90.8%、69.3%、60.0%和62.8%,植物主要包括沿海紫金牛Ardisia japonica、粗叶榕Ficus hirta、枇杷叶紫珠Callicarpa kochiana、杜茎山Maesa japonica、山姜Alpinia japonica、鳞毛蕨Arachniodes hasseltii、狗脊Woodwardia japonica、菝葜Smilax china等。2023年12的林分调查显示:N0、N1、N2和N3处理的林分郁闭度分别为0.86、0.84、0.84和0.83,林分密度分别为1 450、1 267、1 500和1 250株·hm−2,平均胸径分别为26.02、25.39、26.10和26.37 cm。
-
2024年8月,在样地内采用对角线法选取5个采样点。在每个取样点清除表层枯枝落叶后,挖取体积为15 cm×10 cm×10 cm的表层原状土样,置于不锈钢饭盒中(防止挤压)保存。4种处理12块样地共采集60份团聚体样品,用低温保温箱尽快运回实验室。
团聚体分离根据DORNODNIKOV等[17]和WANG等[18]改良的干筛法:样品在低温条件下(4 ℃)风干至土壤含水量达到塑限(含水量约10%~15%),沿着土壤自然脆弱带轻轻掰开,去除可见的根系、石砾等,过8 mm筛。将同一样地的表层团聚体样品混合,合并为12份混合样品。每份样品通过四分法取500 g,用振筛机分离出粗大团聚体(>2.00 mm)、细大团聚体(0.25~2.00 mm)和微团聚体(<0.25 mm)3个粒径。分离后的各粒径组分样品置于−20 ℃冰箱保存。
-
MBC和MBN采用氯仿熏蒸浸提法测定[19]:称取6份过2 mm筛的鲜土12.5 g,3份(不熏蒸)加入50 mL 0.5 mol L−1硫酸钾溶液,振荡30 min后离心过滤;另3份(熏蒸)放入真空干燥器中,在黑暗条件下经氯仿熏蒸24 h后,按相同方法加入硫酸钾溶液浸提、振荡、离心、过滤。滤液使用TOC分析仪(multi N/C
3100 )进行测定。MBP采用氯仿熏蒸浸提法测定[19]:称取3份过2 mm筛的鲜土5.0 g,1份(熏蒸)放入真空干燥器在黑暗条件下用氯仿熏蒸24 h,2份不做熏蒸处理。将熏蒸土样和其中1份未熏蒸土样各加入100 mL 0.5 mol·L−1碳酸氢钠溶液,振荡30 min后离心过滤;1份未熏蒸土样加入0.5 mL 250 mg·L−1 磷酸二氢钾溶液后,再加入100 mL 0.5 mol·L−1 碳酸氢钠溶液,振荡30 min后离心过滤。吸取上述3种提取液10 mL,加入5 mL 5 mg·L−1磷标液,用适量1.0 mol·L−1盐酸溶液中和,放置4 h (期间间隙振荡),再加入4 mL钼锑抗显色剂,定容摇匀,显色30 min后用紫外分光光度计在880 nm波长处进行比色测定。
-
土壤团聚体酶活性使用酶标仪法测定[20]。按照苏州梦犀生物医药科技有限公司提供的β-1, 4-葡萄糖苷酶(BG,碳获取酶)检测试剂盒(M1404A)、N-乙酰-β-D-葡萄糖苷酶(NAG,氮获取酶)检测试剂盒(M1420A)、亮氨酸氨基肽酶(LAP,氮获取酶)检测试剂盒(M1421A)和酸性磷酸酶(AP,磷获取酶)检测试剂盒(M1409A)操作说明,对样品进行前处理后,使用FIexA-200酶标仪进行比色测定,其中BG、NAG和LAP活性在405 nm波长下测定,而AP活性则在660 nm波长下测定。酶碳磷比用BG/AP表示,酶碳氮比用BG/(NAG+LAP)表示,酶氮磷比用(NAG+LAP)/AP表示。
-
采用SPSS 19.0软件,以施氮水平和团聚体粒径为固定效应、标准地为随机效应,构建混合线性模型,分析施氮水平、团聚体粒径及其交互作用对团聚体微生物量碳氮磷、酶活性及其化学计量比的影响。若施氮水平与团聚体粒径存在交互作用,则进行简单效应分析;若无显著交互作用,则移除交互项后重新拟合模型。采用线性回归分析上述指标间的相互关系,显著性水平设定为0.05。运用Origin 2021软件绘制柱状图和回归分析图。
-
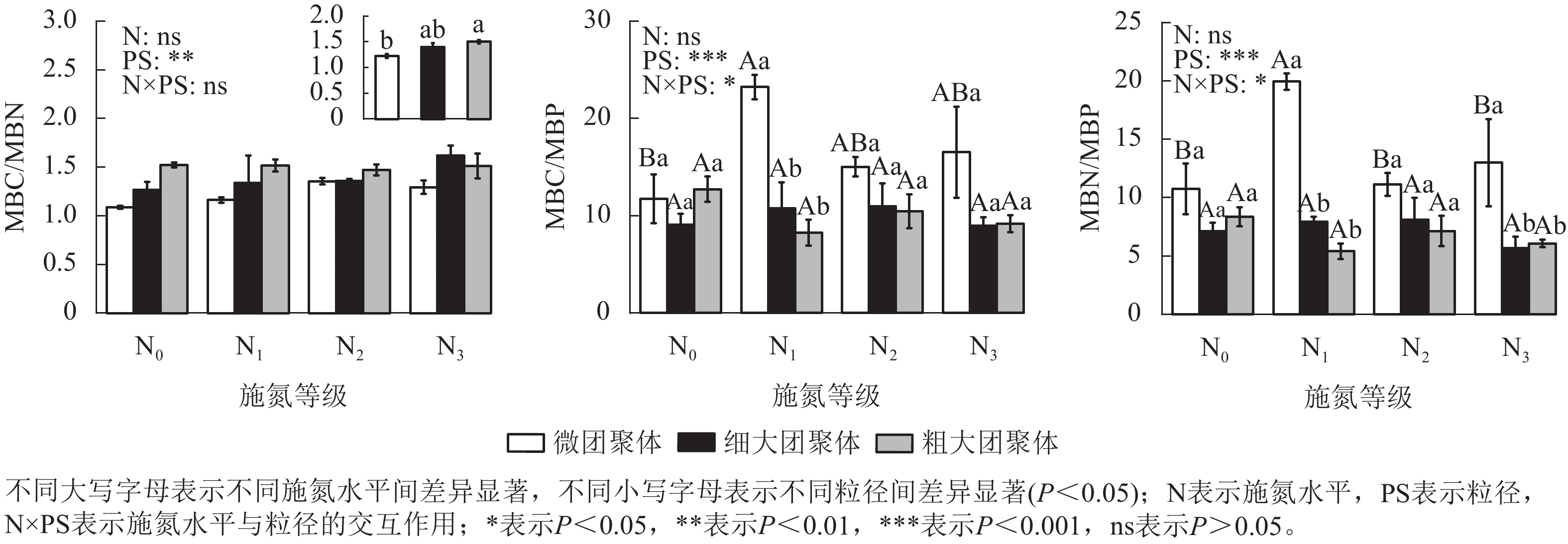
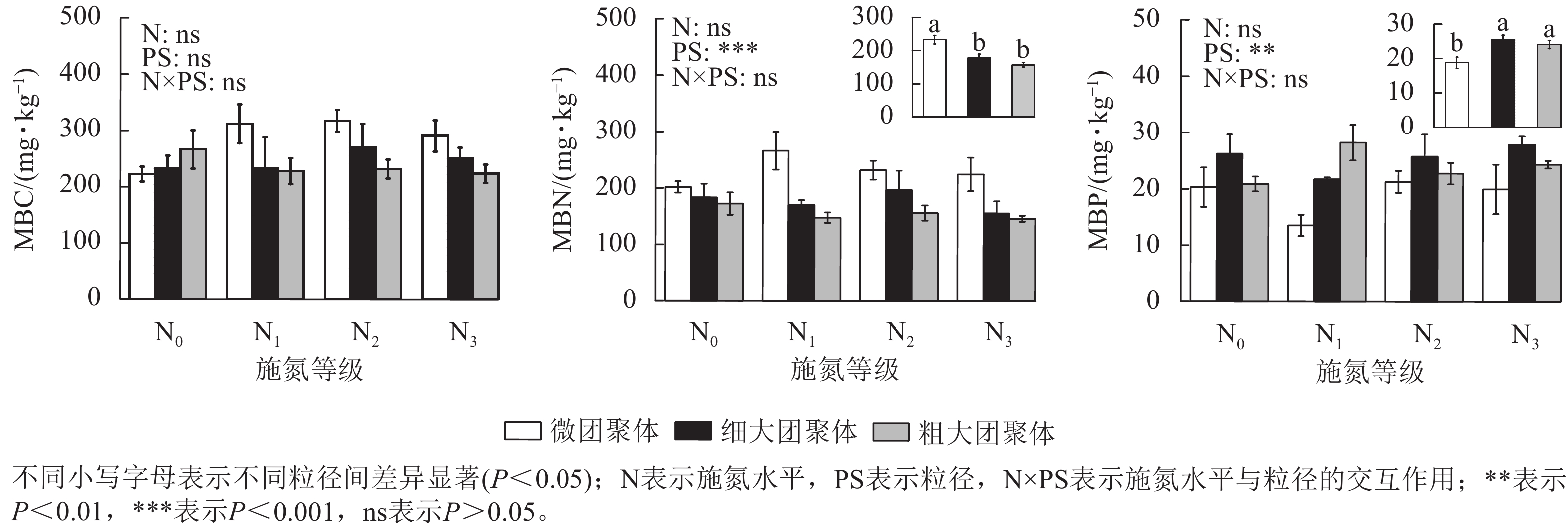
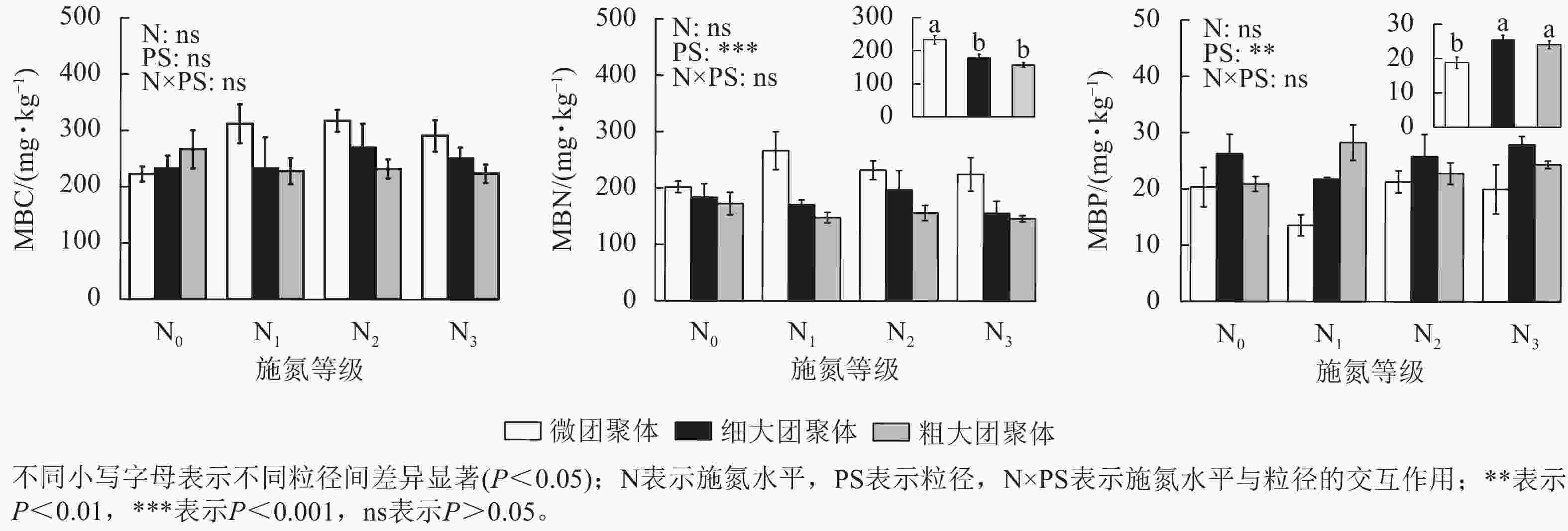
由图1可见:粒径对团聚体MBN和MBP质量分数具有显著影响,而施氮水平、粒径及其交互作用对团聚体MBC无显著影响。细大和粗大团聚体的MBN质量分数比微团聚体分别低23.61%和32.65%(P<0.05),而MBP质量分数则分别高35.31%和28.15%(P<0.05)。
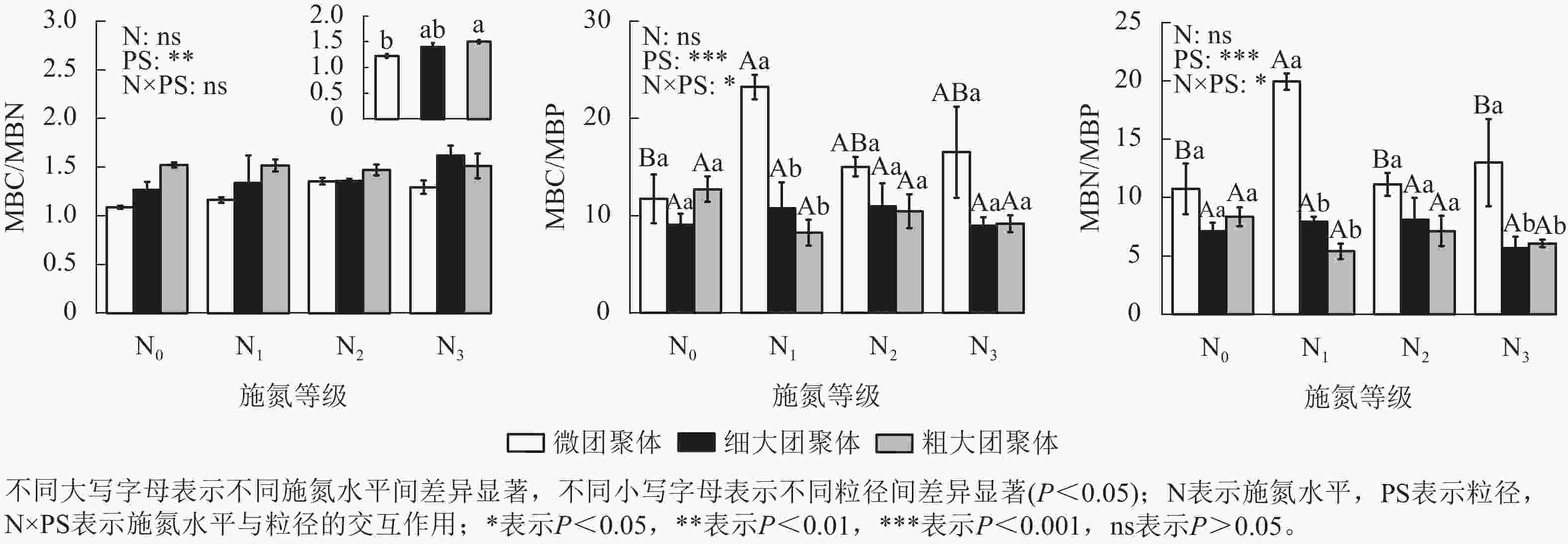
由图2可见:团聚体MBC/MBN仅受粒径的影响,细大和粗大团聚体的MBC/MBN比微团聚体分别高13.87%和22.84%(P<0.05)。粒径、施氮水平与粒径的交互作用对MBC/MBP和MBN/MBP具有显著影响(P<0.05),微团聚体N1处理的MBC/MBP和MBN/MBP分别比N0处理高97.57%和85.51%(P<0.05),细大和粗大团聚体的MBC/MBP和MBN/MBP在各施氮处理间无显著差异。N1处理的细大和粗大团聚体MBC/MBP分别比微团聚体低53.72%和64.43%,MBN/MBP分别低60.25%和72.89%(P<0.05);N3处理的细大和粗大团聚体MBN/MBP分别比微团聚体低56.22%和53.25%;N2处理的MBC/MBP和MBN/MBP在各粒径间差异不显著。
-
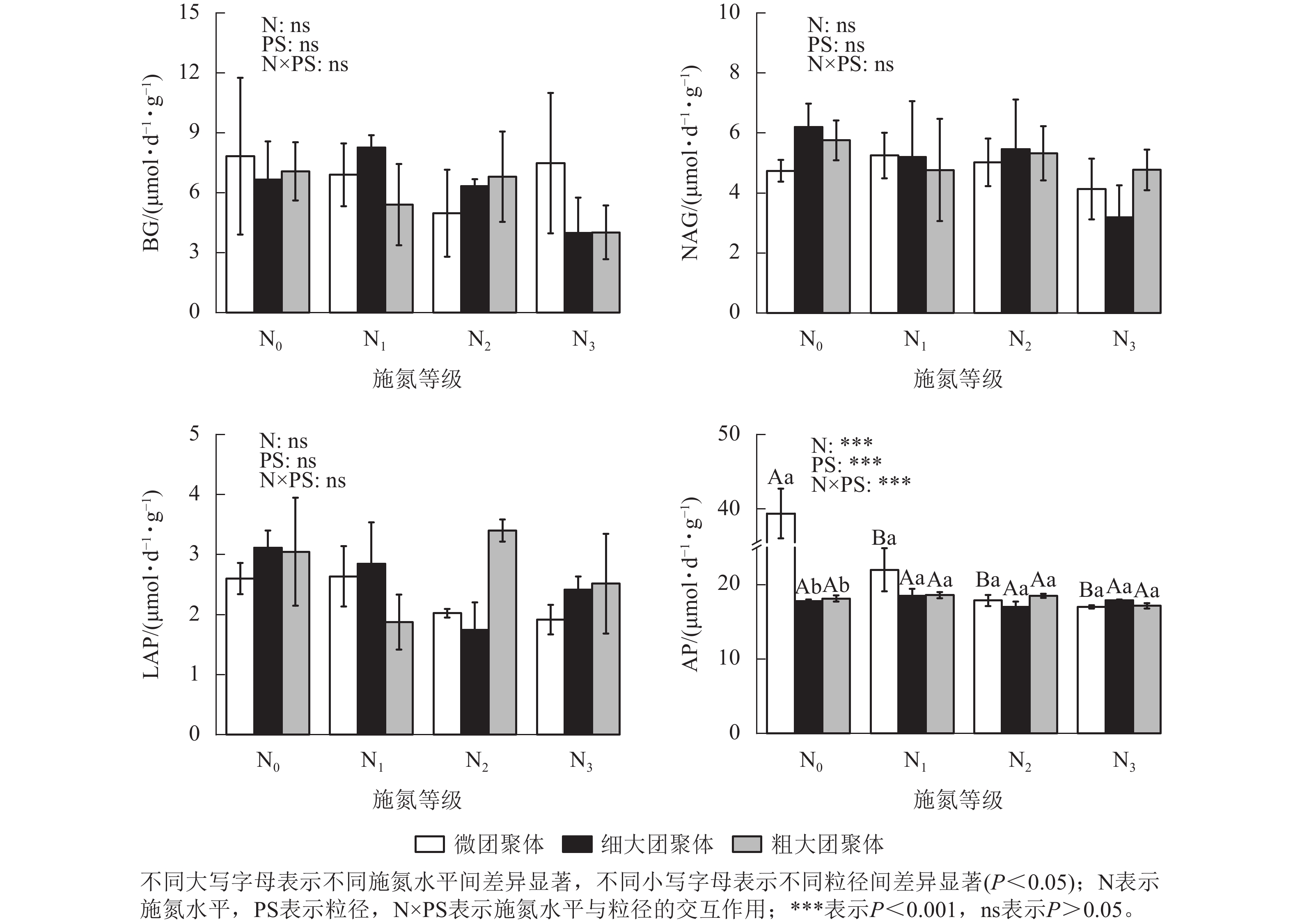
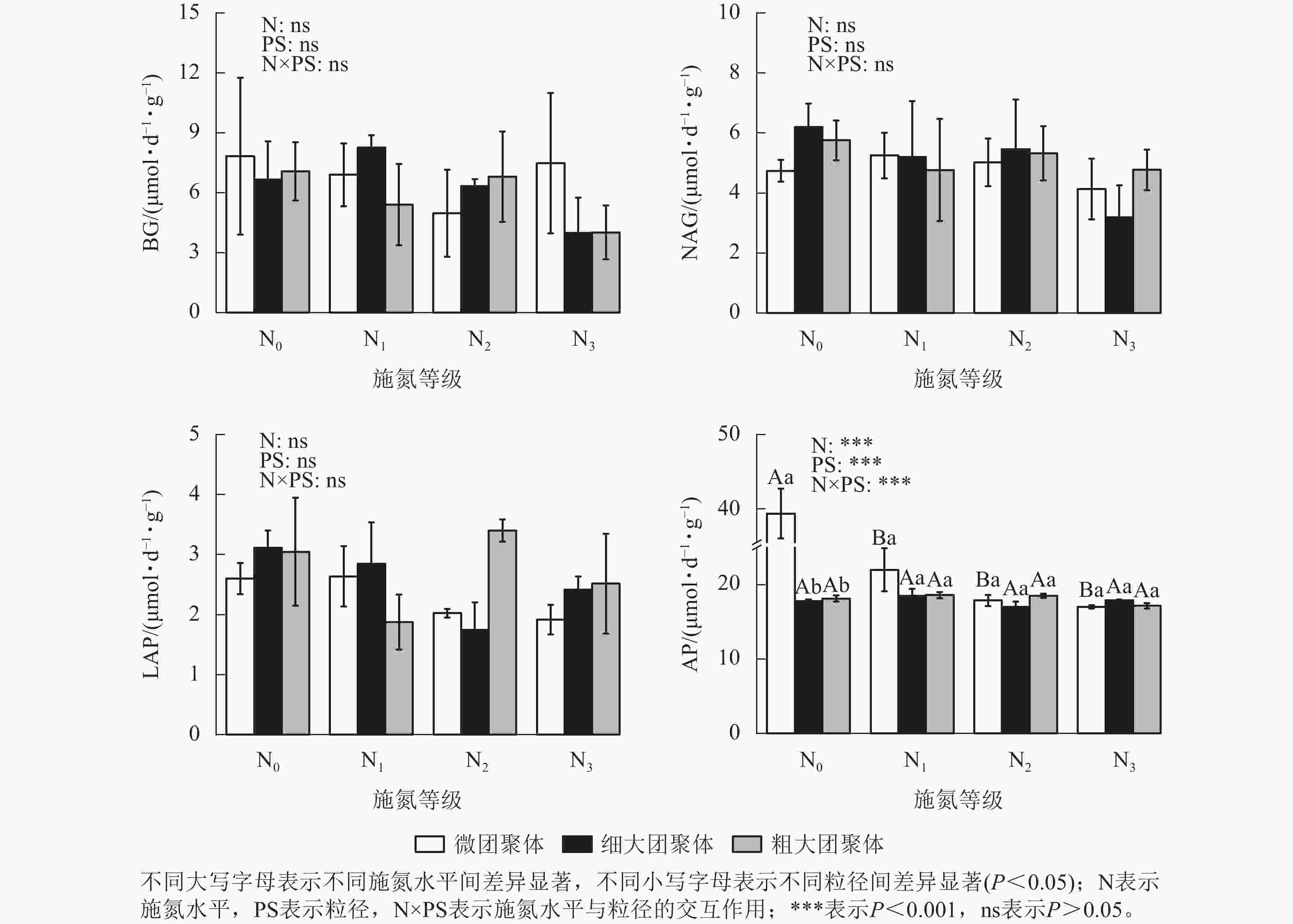
由图3可见:施氮水平、粒径及其交互作用对团聚体BG、NAG和LAP活性均无显著影响,但对AP活性影响显著(P<0.01)。与N0处理相比,微团聚体的N1、N2和N3处理使AP活性分别降低了44.14%、54.60%和56.77%(P<0.05),细大和粗大团聚体的AP活性在各施氮水平间无显著差异。N0处理的细大和粗大团聚体AP活性分别比微团聚体低54.84%和53.92%(P<0.05),其他施氮水平的AP活性在各粒径间差异不显著。
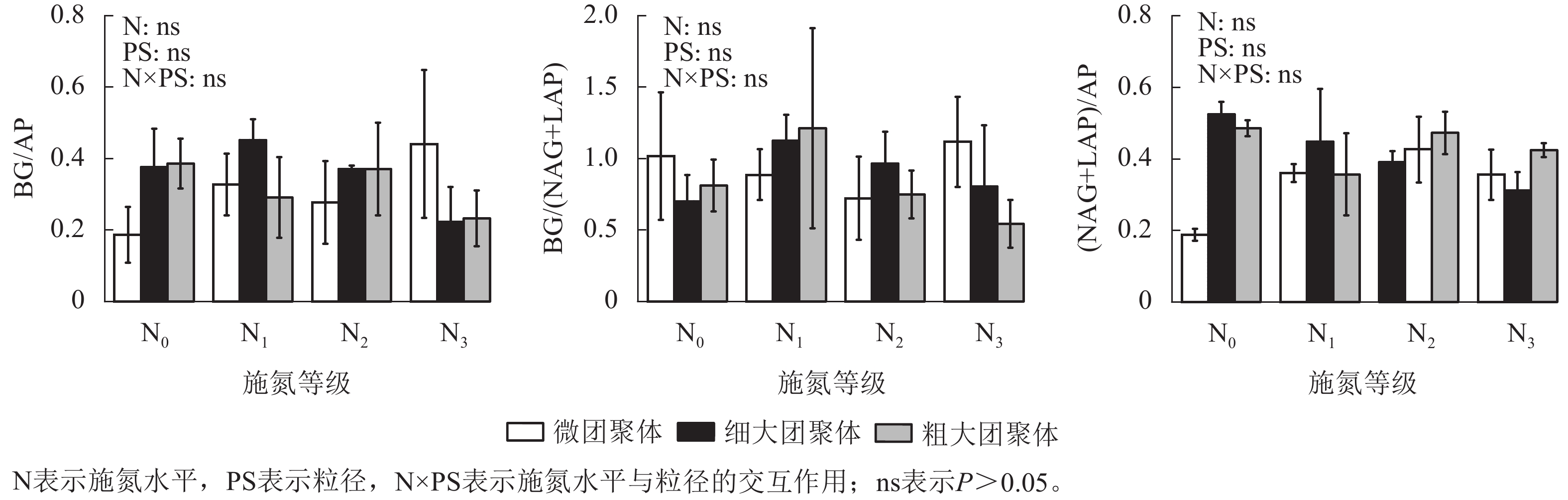
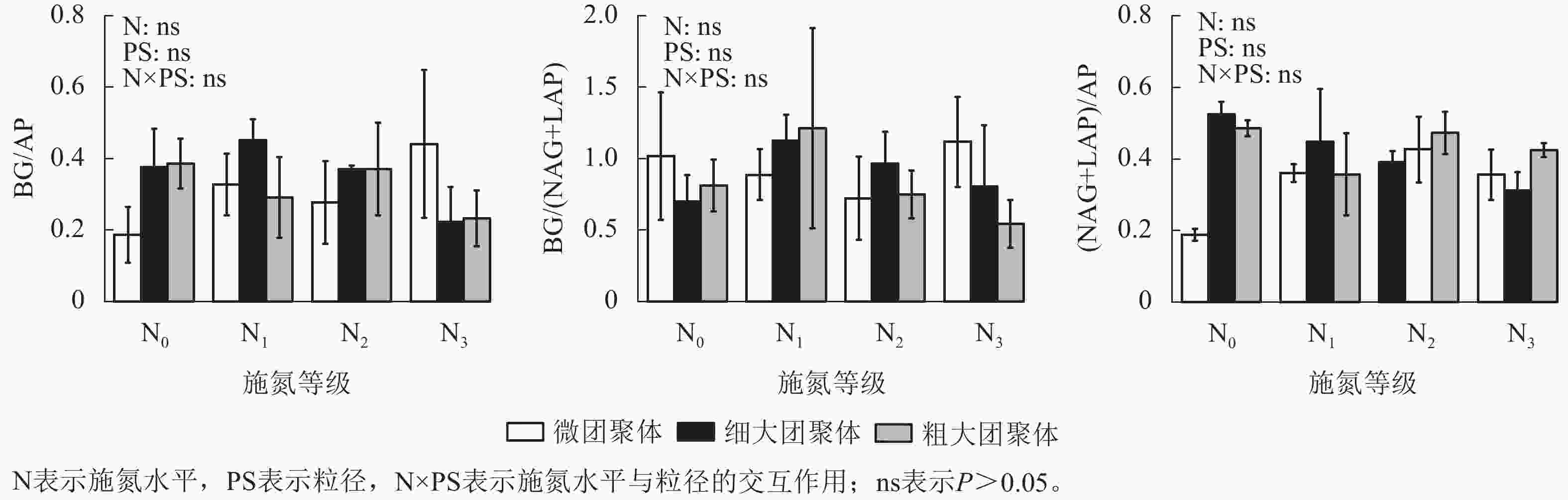
由图4可见:施氮水平、粒径及其交互作用均未显著影响团聚体BG/(NAG+LAP)、BG/AP和(NAG+LAP)/AP,各酶计量比在不同施氮水平和粒级下的变化范围分别为0.54~1.21、0.19~0.45和0.19~0.52。
-
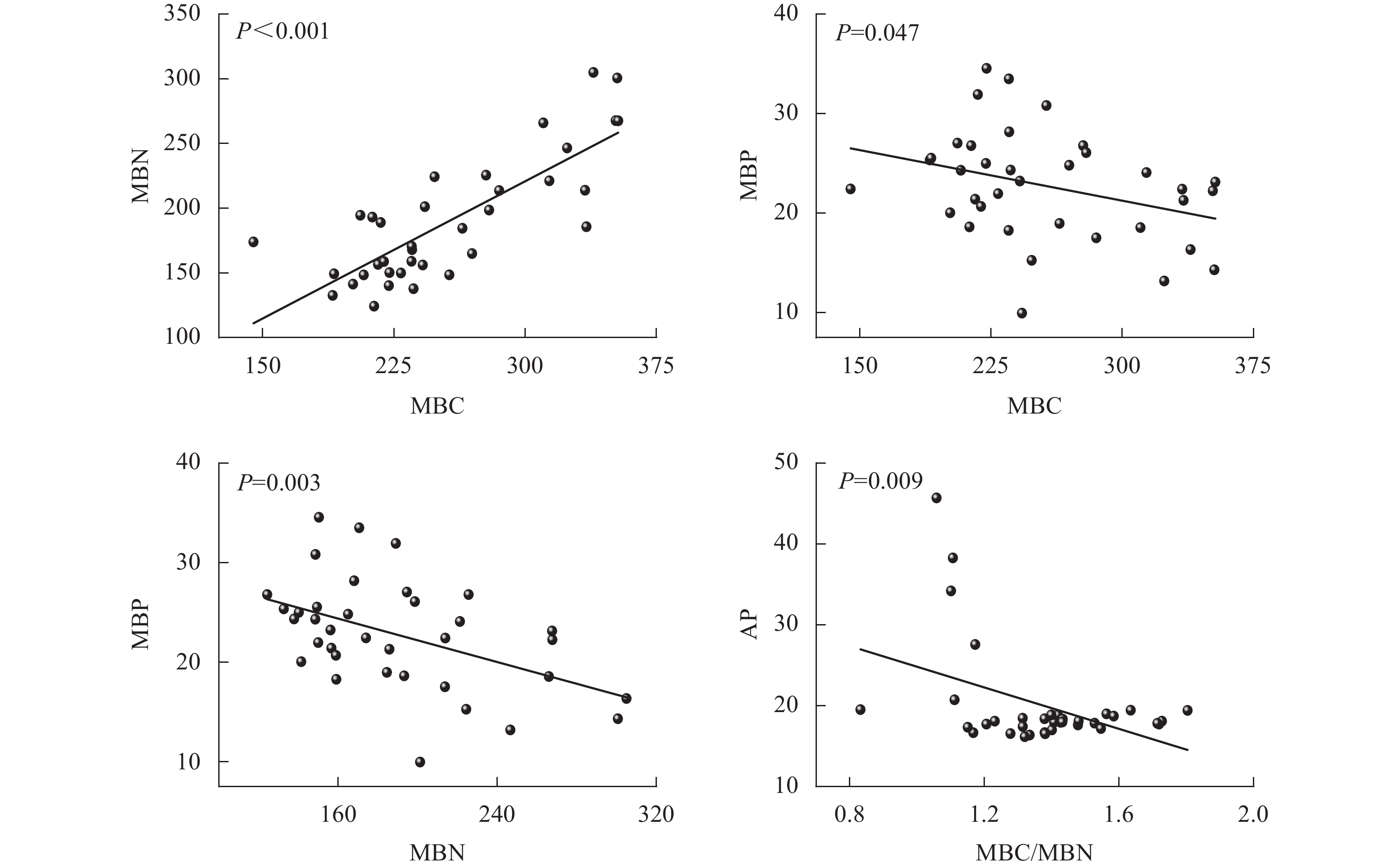
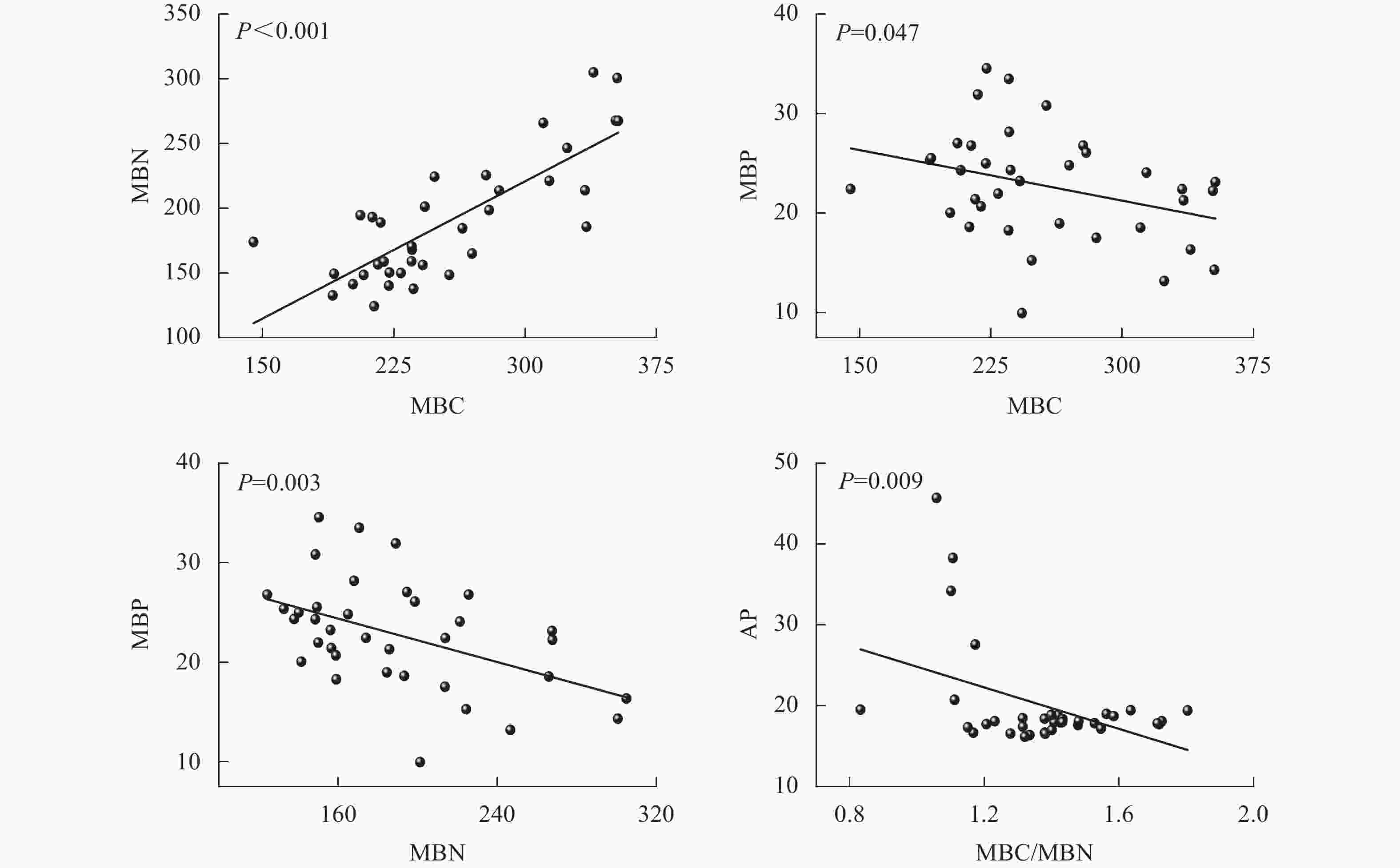
由图5可见:团聚体MBC与MBN呈正相关(P<0.001),而MBC与MBP、MBN与MBP和MBC/MBN与AP呈负相关(P<0.05),其他指标间相关性均不显著。
-
土壤微生物量变化能有效指示碳氮磷循环过程的关键特征[21]。本研究发现:长期氮添加未显著改变团聚体MBC、MBN和MBP含量,这可能是因为氮添加会改变土壤微生物群落结构,而群落通过调整自身代谢途径和功能适应了氮素环境,从而维持了微生物量碳氮磷的相对稳定[22]。MBN含量随团聚体粒径增大而降低,这与周学雅等[23]的研究结果一致。微团聚体因孔隙结构细小,有利于溶解性有机氮的积累,这些有机氮可被微生物直接利用,从而维持较高的MBN含量[24];与之相反,大团聚体孔隙较大,增强了氧气流通性,加速了有机质矿化过程,致MBN含量相对较低[25]。细大和粗大团聚体MBP含量显著高于微团聚体,这可能与不同粒径团聚体对磷的吸附、固定及利用效率有关,大粒径团聚体因磷素有效性提升促进了微生物对磷的吸收利用[26]。
MBC/MBN的大小可表征土壤氮素的供应能力(比值越小,有效性越高),也可反映微生物群落的结构特征(如真菌与细菌的比例)[27]。本研究中细大和粗大团聚体的MBC/MBN显著高于微团聚体,表明大团聚体中真菌相对丰度更高。因为相对于微团聚体,大团聚体的大孔隙结构更有利于真菌菌丝穿透和网络构建,且其包裹的新鲜有机质为真菌提供了更多食物来源[28]。MBC/MBP反映微生物对磷有效性的调节作用,该比值的减小通常意味着微生物磷利用效率的提高和磷释放潜力的增加,最终促进土壤有效磷的供给[29];而MBN/MBP则反映植物对氮、磷养分的相对需求强度[30]。本研究发现,N1施氮水平下增加了微团聚体的MBC/MBP和MBN/MBP,说明氮添加使植物对磷素的相对需求超过氮素。另外,微团聚体的MBC/MBP和MBN/MBP相对较高,表明其磷供给潜力低于细大和粗大团聚体,这是因为大团聚体结构疏松、稳定性低,易受水热条件波动而分解,有助于释放其固持的磷素;而微团聚体尽管可能富集有效磷,但因其致密结构限制磷素释放,导致实际供给能力受限[31]。这种差异揭示了长期氮添加通过重构团聚体结构,驱动微生物养分分配策略的转变,表现为大团聚体中微生物增强磷捕获能力(低MBC/MBP),同时减少氮储存(低MBN/MBP),而微团聚体则维持高磷固持策略。
-
土壤水解酶可通过催化高分子及难降解有机物的水解反应,将其转化为生物可利用养分,进而驱动生态系统的物质循环与能量流动[32]。本研究发现:长期氮添加仅改变了团聚体中磷获取酶活性(如AP酶),而碳、氮获取酶活性无显著变化,表明碳氮获取酶活性受氮添加的影响较小。长期氮输入可能会加剧土壤磷限制,促使植物和微生物通过分泌更多的AP来调控磷的有效性[33−34]。然而,施氮后,微团聚体的AP活性显著降低,这可能是因为长期氮添加会酸化土壤,加速矿物态磷的溶解,提高团聚体内磷有效性,导致微生物减少对AP合成的资源投入[35]。大团聚体AP活性对施氮无显著响应,其敏感性低于微团聚体。
在未施氮(N0)条件下,微团聚体AP活性显著高于细大和粗大团聚体,表明小团聚体具有更强的有机磷矿化能力,有利于提高土壤养分循环效率。这种粒径差异主要源于3个方面:一是小团聚体通过有机-无机复合体更易吸附并固定酶蛋白,提供物理保护,减少酶降解;二是小团聚体富集高丰度细菌群落(主要酶生产者),其多样性优势促进了酶的合成与积累[36];三是磷素在团聚体粒径间的异质性分布直接调控酶活性表达[9]。而大团聚体因孔隙较大,稳定性较低,其酶活性易受外界环境变化的影响 [31, 37],进一步加剧了与小团聚体的酶活性差距。长期施氮下,AP活性在团聚体各粒径间差异均不显著,可能是由于氮输入降低了土壤pH,改变了微生物群落结构,抑制了微生物分泌酶活性,导致酶活性在粒径间响应趋同[14]。
土壤酶化学计量比可反映土壤养分资源的有效性和微生物对养分需求的变化[38]。本研究中,不同氮添加水平下团聚体酶化学计量比均未呈现显著差异,表明该区域微生物对碳、氮、磷获取酶的分泌策略在长期氮输入压力下保持稳定。值得注意的是,团聚体酶碳磷比和酶氮磷比均值较低,低于全球尺度水平0.62和0.44[39],说明本区域内土壤微生物对磷获取酶的投资高于碳氮获取酶,暗示微生物的生长可能受磷素限制,这与亚热带生态系统生产力普遍受磷限制的结论相吻合[40]。
-
本研究发现,土壤团聚体MBC与MBN正相关。CLEVELAND等[41]指出,碳和氮在微生物体内具有相对稳定的比例关系,因为碳是微生物生长的能量来源,氮是蛋白质、核酸等主要成分,两者在微生物细胞合成中是紧密关联的。而MBC、MBN与MBP则呈现负相关,当土壤有效磷不足时,微生物增加多聚磷酸盐的储存(MBP增加),同时减少蛋白质和核酸的合成(MBC、MBN降低),这与陈露等[42]对湖北麦区磷肥减施后的研究结果类似。团聚体MBC/MBN与AP活性的负相关也说明在磷限制时,微生物可能倾向于减少生物量积累(较低的MBC/MBN),将更多的资源用于磷酸酶的合成,从而提升磷获取效率,这体现了微生物资源分配的权衡策略。
-
微团聚体对氮添加最敏感,其微生物量碳磷比和氮磷比在N1处理下显著升高,酸性磷酸酶活性显著降低,而细大和粗大团聚体的微生物量碳氮磷和酶活性对氮添加响应不显著。团聚体酶碳磷比和酶氮磷比低于全球水平,证实了亚热带生态系统的磷限制特征。团聚体微生物量碳氮比与酸性磷酸酶负相关,体现了微生物资源分配的权衡策略。本研究强调了土壤团聚体大小在调节微生物对氮沉降反应中的重要性,应将其纳入生态系统模型,以预测全球变化条件下的土壤生物地球化学循环。
Effects of long-term nitrogen addition on microbial biomass carbon, nitrogen, and phosphorus, enzyme activities, and their stoichiometric characteristics in soil aggregates of Cunninghamia lanceolata plantations
doi: 10.11833/j.issn.2095-0756.20250354
- Received Date: 2025-06-27
- Rev Recd Date: 2025-12-14
-
Key words:
- soil aggregate /
- microbial biomass /
- enzyme activity /
- nitrogen addition /
- Chinese fir plantation
Abstract:
| Citation: | YAN Wenjin, ZHU Liqin, LI Jingkai, et al. Effects of long-term nitrogen addition on microbial biomass carbon, nitrogen, and phosphorus, enzyme activities, and their stoichiometric characteristics in soil aggregates of Cunninghamia lanceolata plantations[J]. Journal of Zhejiang A&F University, 2025, 43(X): 1−10 doi: 10.11833/j.issn.2095-0756.20250354 |









 DownLoad:
DownLoad: