-
小麦Triticum aestivum作为全球重要的粮食作物之一,其生长发育状况、产量和品质与粮食安全息息相关。受气候变化的影响,全球约3/4的陆地呈现干旱化趋势,持续干旱胁迫导致小麦减产,这给农业生产带来了巨大的损失。中国干旱与半干旱地区的总面积占全国土地总面积的52.5%,区域内干旱灾害频发,对小麦生产构成严重威胁[1]。而种子萌发是小麦苗期建成的关键阶段,干旱胁迫下水分亏缺易造成田间缺苗断垄的现象,进而影响产量[2−4]。因此,探寻有效调控措施缓解干旱胁迫对小麦萌发期的抑制作用,对保障小麦高产稳产具有重要意义。近年来,通过添加外源化学物质调控作物萌发期抗旱能力研究较多,如水杨酸[5−6]、褪黑素[7]、黄腐酸[8]和多胺[9−10]等浸种方式,因操作简便有效而备受关注。多胺(PAs)是一类广泛存在于生物体内、具有较强生理活性的低分子量脂肪族含氮化合物,它不仅影响植物的正常生长发育,还与非生物抗逆胁迫关系密切[11−12],因此近年来常作为抗逆生长调节剂被广泛应用。常见的多胺有四胺精胺(Spm)、三胺亚精胺(Spd)、二胺腐胺(Put)和尸胺(Cad)等。其中,具有多价阳离子特性的精胺和亚精胺由于生理功能更强,在维持植物抗旱性方面发挥着不可替代的作用[13]。外源多胺在一定程度上能够缓解植物干旱胁迫下受到的伤害。如外源精胺可通过提高酶活性、光合色素含量、降低丙二醛含量来减少水分亏缺的不利影响[14];外源亚精胺可减轻干旱胁迫对玉米Zea mays的生长、光合结构和功能的损害[15];外源亚精胺可显著缓解干旱胁迫对冬青Ilex chinensis幼苗的抑制作用[16]。同外源多胺对胁迫的缓解作用效果不同,姜惠丽等[17]和王素平等[18]在研究3种多胺缓解盐胁迫对黄瓜Cucumis sativus幼苗生长抑制以及影响离子吸收与运输的作用时发现:其缓解效果从大到小依次为亚精胺、精胺、腐胺。目前,关于多胺对小麦抵御干旱胁迫的研究亦有相关报道,但外源精胺和亚精胺对不同抗旱性品种小麦萌发的生理调节机制研究较少。鉴于此,本研究以干旱适应性不同的2种小麦品种为材料,研究不同浓度精胺、亚精胺浸种对聚乙二醇(PEG-6000)模拟干旱胁迫下小麦萌发期生长及生理指标的影响,筛选最优浓度与关键抗旱指标,以期为小麦抗旱生产和粮食安全保障提供科学依据。
-
以干旱适应性不同的2个小麦品种[‘百农201’‘Bainong 201’(抗旱性较弱)和‘矮抗58’‘Aikang 58’(抗旱性较强)]为材料,该小麦品种由西北农林科技大学分子染色体工程实验室提供;试验所用试剂PEG-6000、精胺和亚精胺均为Sigma公司产品。
-
随机挑选‘百农201’和‘矮抗58’无病虫害且完整的种子,用体积分数为5%的次氯酸钠表面消毒15 min,无菌蒸馏水冲洗3~5次。对照组种子用蒸馏水浸种,各试验组种子用不同浓度(0.05、0.10和0.20 mmol·L−1)精胺和亚精胺浸种12 h备用。将处理好的种子放置于铺有双层滤纸的干净培养皿中,加入处理液浸没过种子并使种子腹沟朝下,且保证每天定时更换处理液以保证其浓度不变,整个培养过程处在12 h光照/12 h黑暗环境中,其他条件均适宜。对照组和试验组精胺、亚精胺浸种处理的‘百农201’和‘矮抗58’如表1所示,每个培养皿放50粒种子,每组处理进行3次重复。
处理
编号精胺组 亚精胺组 精胺浓度/
(mmol·L−1)PEG-6000
质量浓度/%亚精胺浓度/
(mmol·L−1)PEG-6000
质量浓度/%1 0 0 0 0 2 0 5 0 5 3 0 10 0 10 4 0 15 0 15 5 0.05 5 0.05 5 6 0.05 10 0.05 10 7 0.05 15 0.05 15 8 0.10 5 0.10 5 9 0.10 10 0.10 10 10 0.10 15 0.10 15 11 0.20 5 0.20 5 12 0.20 10 0.20 10 13 0.20 15 0.20 15 说明:处理编号1为对照组(ck),蒸馏水浸种,无PEG-6000干旱胁迫;处理编号2~4为PEG-6000胁迫处理;处理编号5~13为精胺/亚精胺浸种后用PEG-6000胁迫处理。 Table 1. Test treatment
-
萌发指标的测定包括种子发芽率、根长和芽长。每天定时观察并记录各处理小麦种子的萌发数量,以胚芽长度达该种子长度的1/2作为种子萌发标准,连续3 d无种子萌发视为萌发结束。发芽结束后,每处理随机挑选10株幼苗,测量根长和芽长。叶绿素(Chl)质量分数、丙二醛(MDA)质量摩尔浓度、脯氨酸(Pro)质量分数和过氧化物酶(POD)活性的测定分别采用比色法、硫代巴比妥酸法、磺基水杨酸法和紫外吸收法。以上具体测定步骤均参照张志良等[19]的方法。
-
运用模糊隶属函数法筛选对小麦幼苗干旱缓解效果最好的处理组,具体方法参照胡胜男等[20]的方法。
-
利用Excel 2010整理数据,使用SPSS 26.0进行差异显著性分析(P<0.05),利用Origin Pro 2022软件作图,采用模糊隶属函数法筛选最佳缓解干旱胁迫的外源精胺和亚精胺浓度。
-
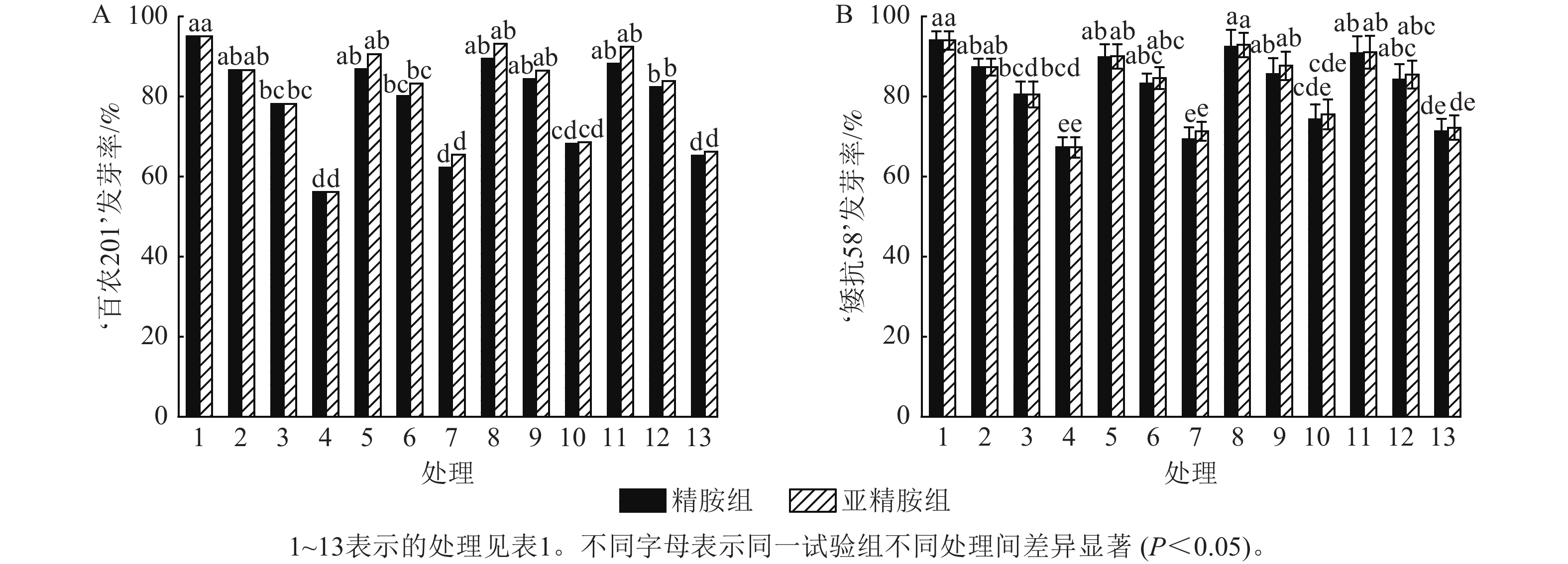
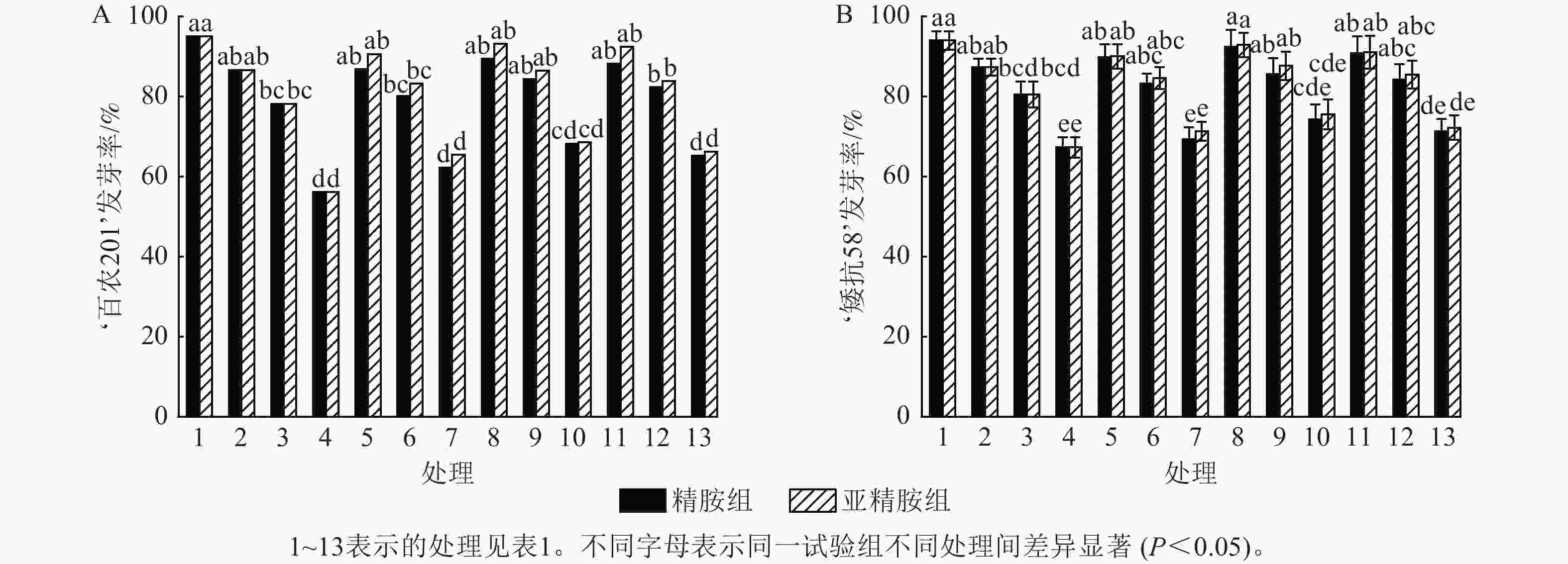
如图1所示:PEG-6000模拟的干旱胁迫抑制了小麦‘百农201’和‘矮抗58’的种子萌发,且发芽率随着PEG-6000质量浓度的升高呈显著下降趋势(P<0.05)。在PEG-6000质量浓度为5%、10%和15%的干旱胁迫下,‘百农201’的发芽率分别较对照下降8.81%、17.72%和40.92% (图1A),‘矮抗58’分别下降7.10%、14.30%和28.40% (图1B),表明抗旱性较弱的‘百农201’种子萌发对干旱胁迫相对更敏感。精胺和亚精胺浸种对抗旱性较强品种‘矮抗58’在干旱胁迫下的发芽率无显著效果,而亚精胺浸种对抗旱性较弱的品种‘百农201’干旱胁迫的发芽率有促进作用,且0.10 mmol·L−1亚精胺浓度对高质量浓度PEG-6000 (15%)的胁迫效果显著(P<0.05)。
-
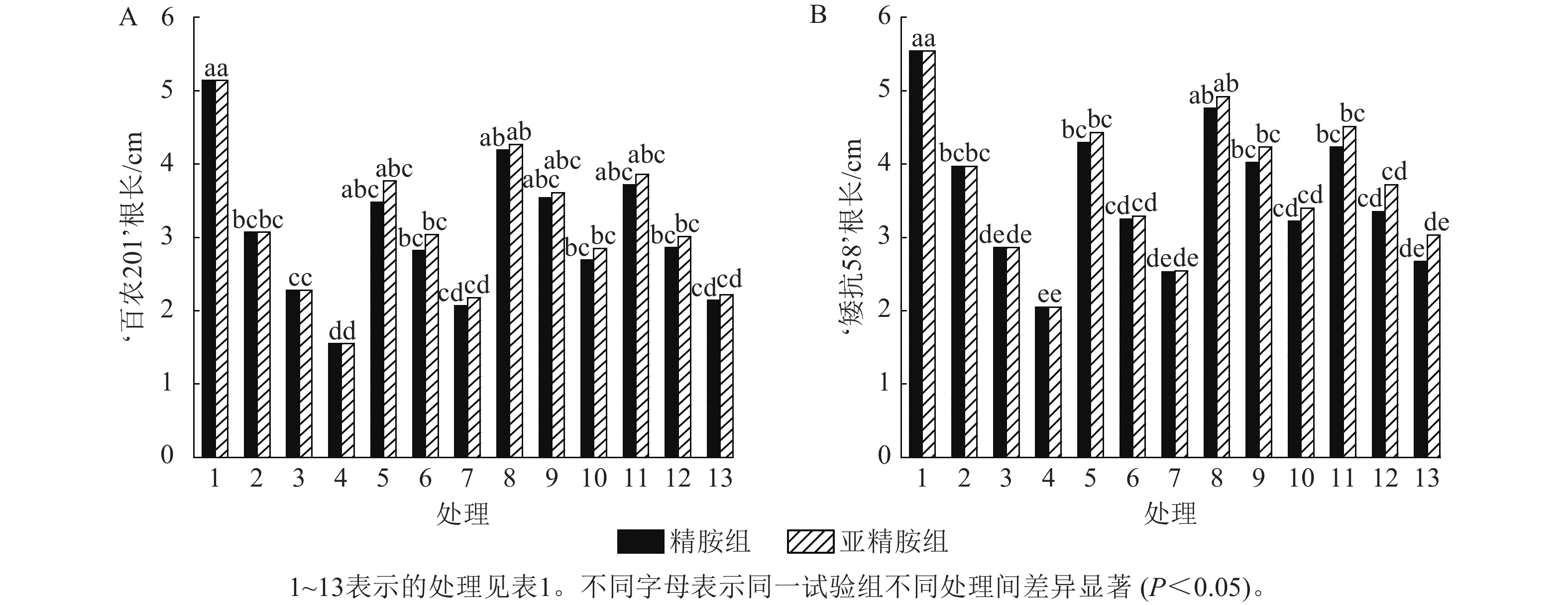
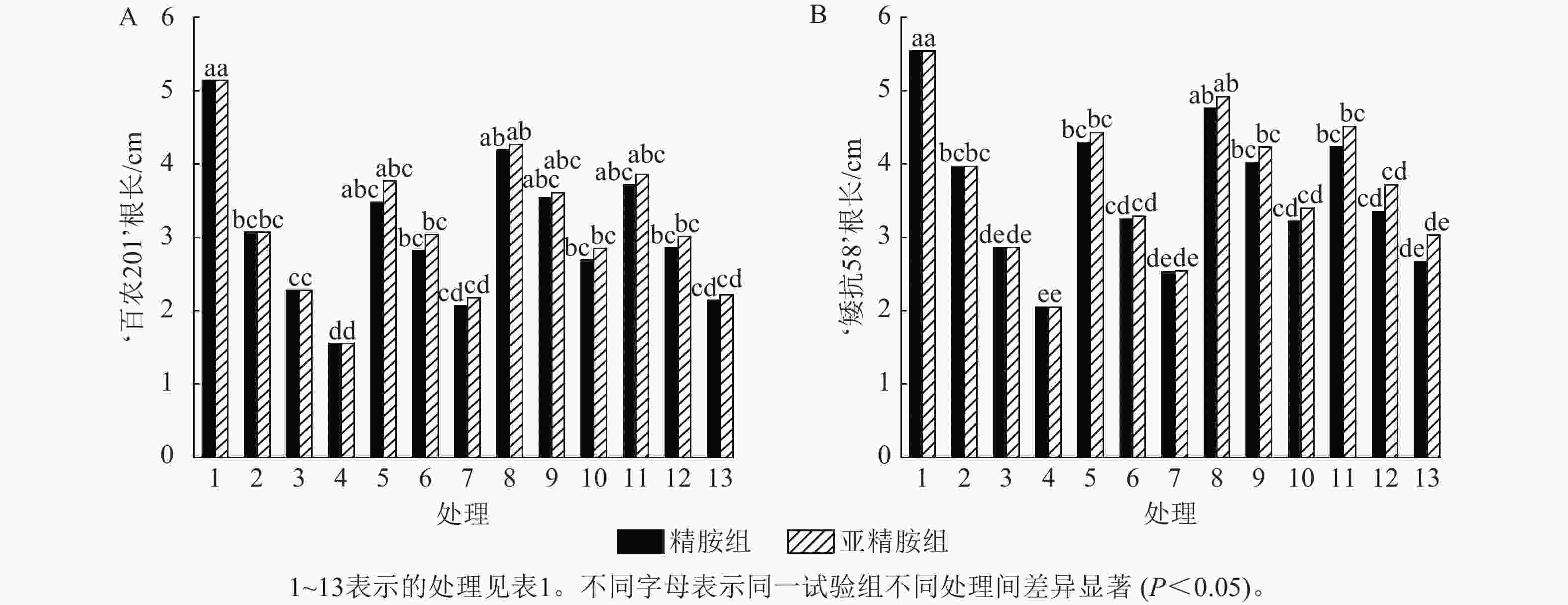
由图2可知:PEG-6000模拟的干旱胁迫抑制了小麦‘百农201’和‘矮抗58’的根长生长,且抑制程度随着胁迫强度的升高而增强。在PEG-6000质量浓度为5%、10%和15%的干旱胁迫下,‘百农201’根长较对照分别下降40.27%、55.64%和69.84%,‘矮抗58’分别下降28.34%、48.38%和63.00%,表明抗旱性较弱的‘百农201’根长对干旱胁迫更敏感。0.10 mmol·L−1精胺浸种后,‘百农201’在质量浓度为15%PEG-6000胁迫下的根长较对应干旱胁迫下显著增长73.55% (P<0.05),说明0.10 mmol·L−1精胺浸种对高质量浓度干旱胁迫下‘百农201’根长的缓解效果更佳。0.10 mmol·L−1精胺浸种后,‘矮抗58’在质量浓度为10%和15%PEG-6000胁迫下的根长较对应干旱胁迫下分别显著增长40.56%和57.07% (P<0.05),说明0.10 mmol·L−1精胺浸种,对中高质量浓度(10%和15%)干旱胁迫下‘矮抗58’根长的缓解效果更佳。亚精胺浸种后,‘百农201’和‘矮抗58’的根长较对应质量浓度PEG-6000干旱胁迫下的根长均有所增加。其中,0.10和0.20 mmol·L−1亚精胺浸种后,在质量浓度为15% PEG-6000干旱胁迫下,‘百农201’的根长较对应干旱胁迫下分别显著增加83.87%和43.23% (P<0.05);0.10 mmol·L−1亚精胺浸种后,在质量浓度为10%和15%PEG-6000干旱胁迫下,‘矮抗58’的根长较对应干旱胁迫下显著提高47.90%和65.85% (P<0.05);在0.20 mmol·L−1亚精胺浸种后,在质量浓度为15%PEG-6000干旱胁迫下,‘矮抗58’的根长较对应干旱胁迫下显著提高47.80% (P<0.05)。说明适宜浓度的精胺和亚精胺浸种可促进干旱胁迫下根长的生长,且0.10 mmol·L−1亚精胺浸种效果更优。
-
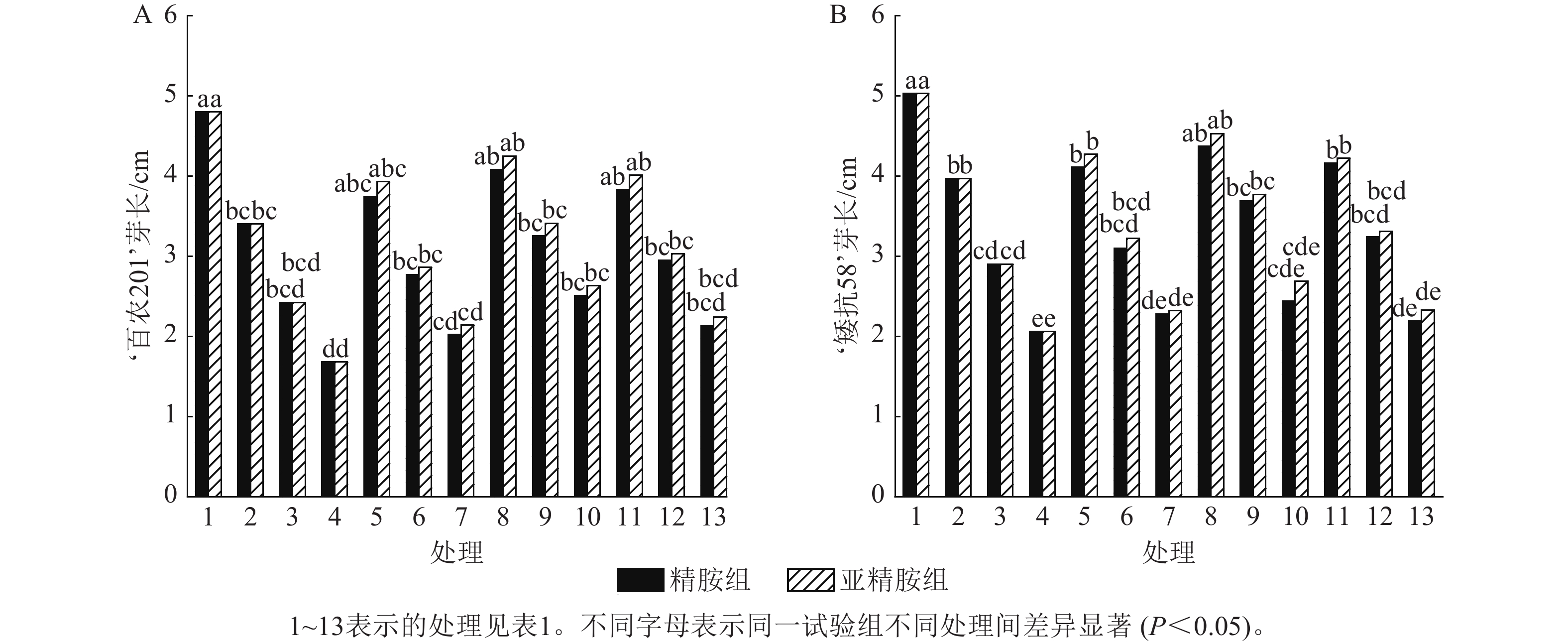
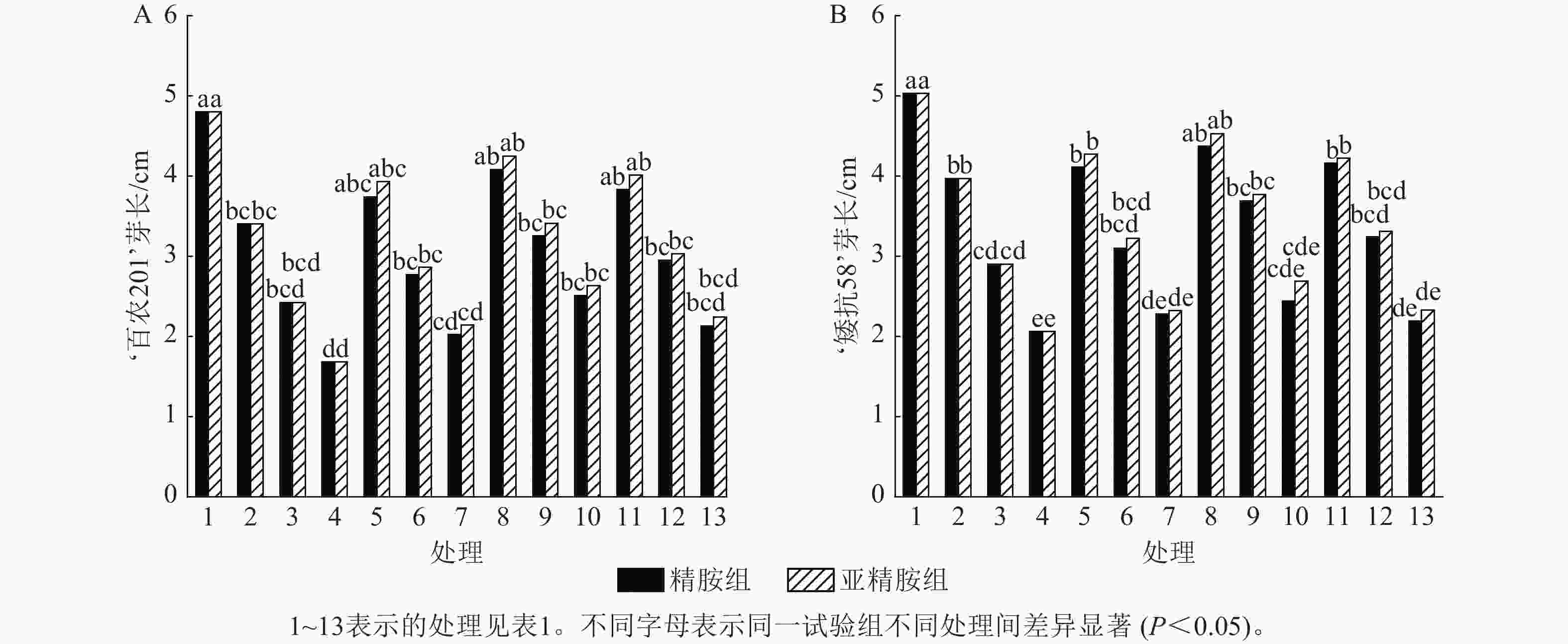
从图3可知:PEG-6000模拟的干旱胁迫抑制小麦‘百农201’和‘矮抗58’芽长的生长。‘百农201’在质量浓度为5%、10%和15% PEG-6000干旱胁迫下,‘百农201’芽长较对照分别下降29.17%、49.58%和65.00%,‘矮抗58’分别下降21.07%、42.35%和59.05%,表明抗旱性较弱的‘百农201’芽长对干旱胁迫更敏感。3种不同浓度的精胺和亚精胺浸种后,‘百农201’和‘矮抗58’芽长较对应浓度PEG-6000胁迫下均有所增加。在质量浓度为15%PEG-6000干旱胁迫下,‘百农201’芽长在0.10 mmol·L−1精胺和亚精胺浸种后较对应浓度干旱胁迫下的芽长分别显著提高49.40%和56.55% (P<0.05);矮抗58’芽长较对应浓度干旱胁迫下均无显著差异。综上说明0.10 mmol·L−1精胺和亚精胺浸种可缓解高质量浓度干旱胁迫对‘百农201’芽长的抑制作用,且亚精胺的缓解效果优于精胺。
-
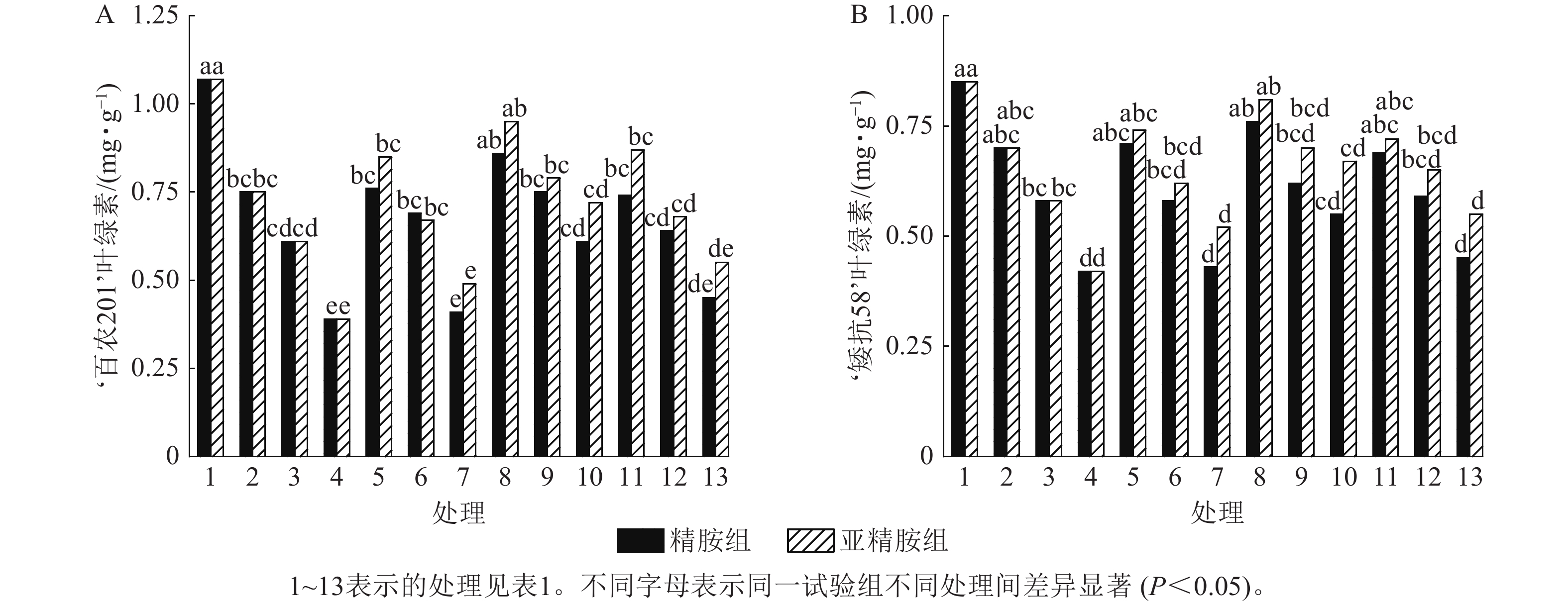
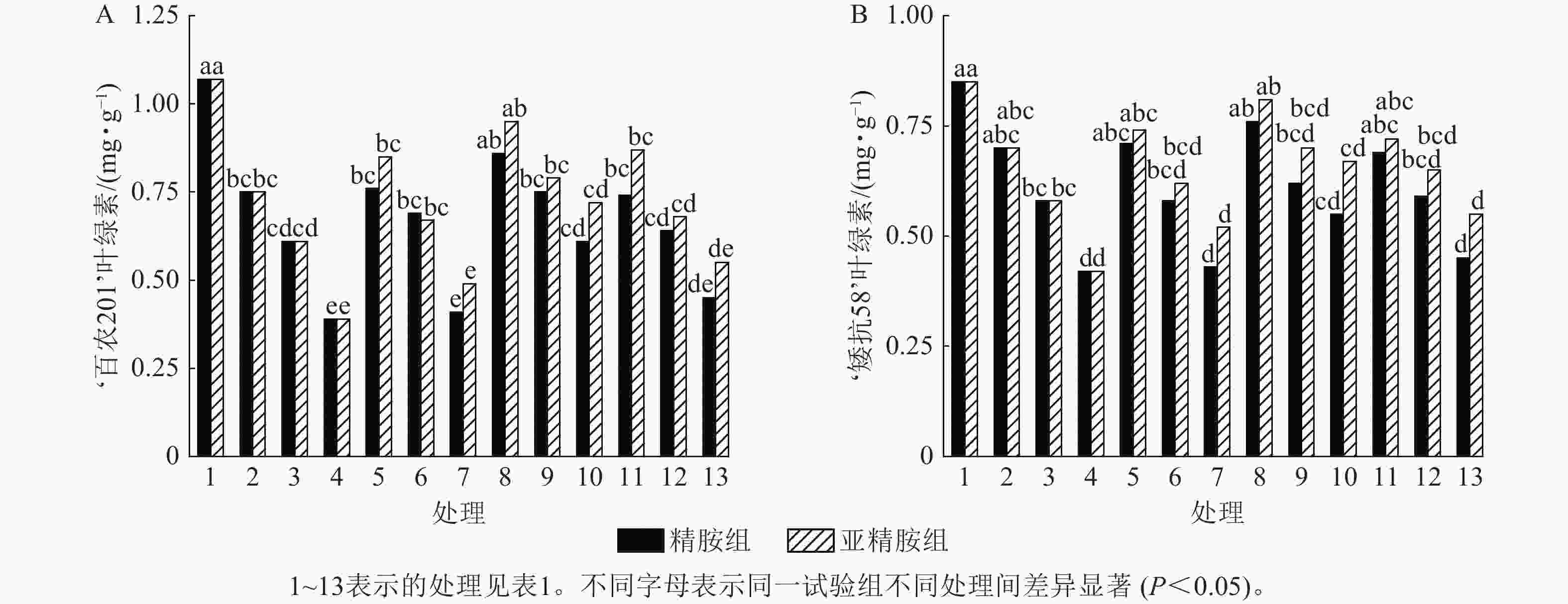
由图4可知:干旱胁迫抑制了小麦‘百农201’和‘矮抗58’叶绿素的产生,且叶绿素质量分数随着PEG-6000质量浓度的升高呈显著下降趋势(P<0.05)。在PEG-6000质量浓度为5%、10%和15%的干旱胁迫下,‘百农201’叶绿素质量分数较对照分别下降29.91%、42.99%和63.55%,‘矮抗58’分别下降17.65%、31.76%和50.59%,表明抗旱性较弱的‘百农201’对干旱胁迫更敏感。0.10 mmol·L−1精胺浸种后,在PEG-6000质量浓度为15%干旱胁迫下,‘百农201’叶绿素质量分数较对应干旱胁迫下的叶绿素质量分数显著提高56.41% (P<0.05);‘矮抗58’在不同浓度精胺浸种后的叶绿素质量分数差异不显著。0.10 mmol·L−1亚精胺浸种后,在质量浓度为15%PEG-6000胁迫下,‘百农201’和‘矮抗58’叶绿素质量分数分别较对应干旱胁迫下显著提高84.62%和59.52% (P<0.05)。说明0.10 mmol·L−1精胺和亚精胺浸种可促进干旱胁迫下叶绿素的产生,其中0.10 mmol·L−1亚精胺浸种对高质量浓度干旱胁迫下的叶绿素产生效果缓解更佳,且对抗旱性较弱的‘百农201’具有更大的缓解调节效应。
-
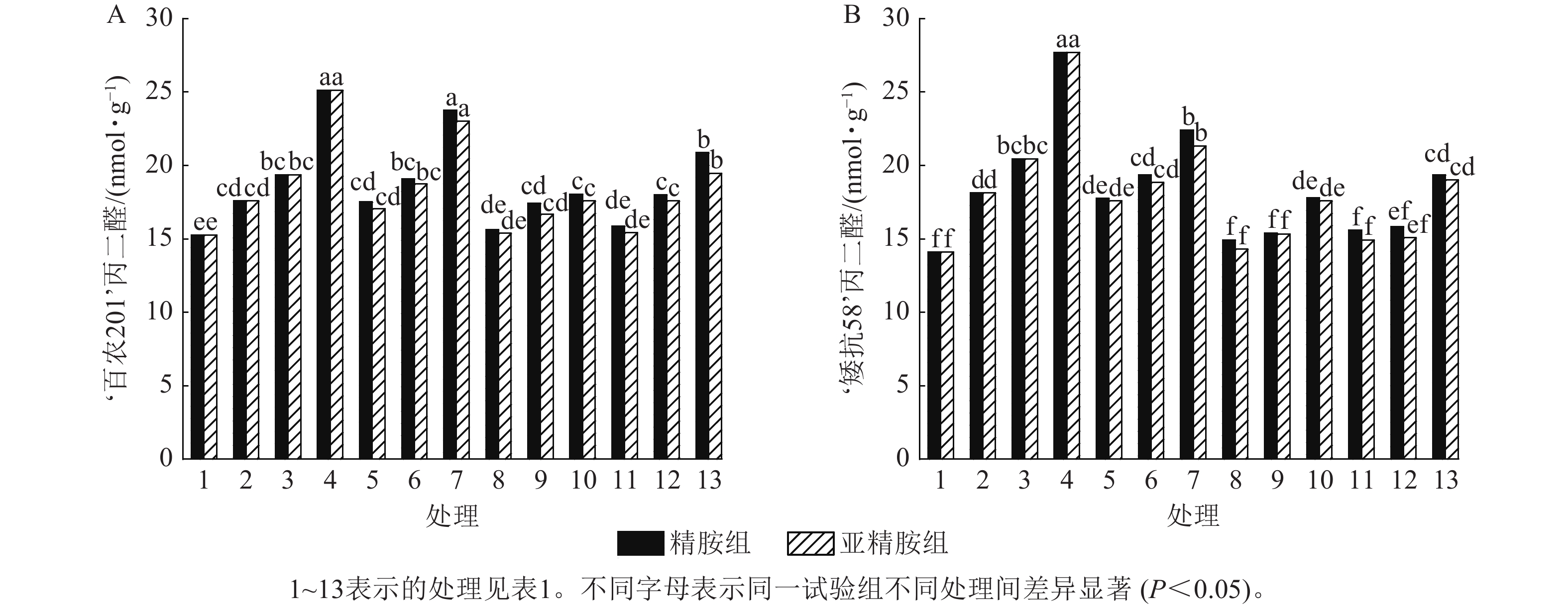
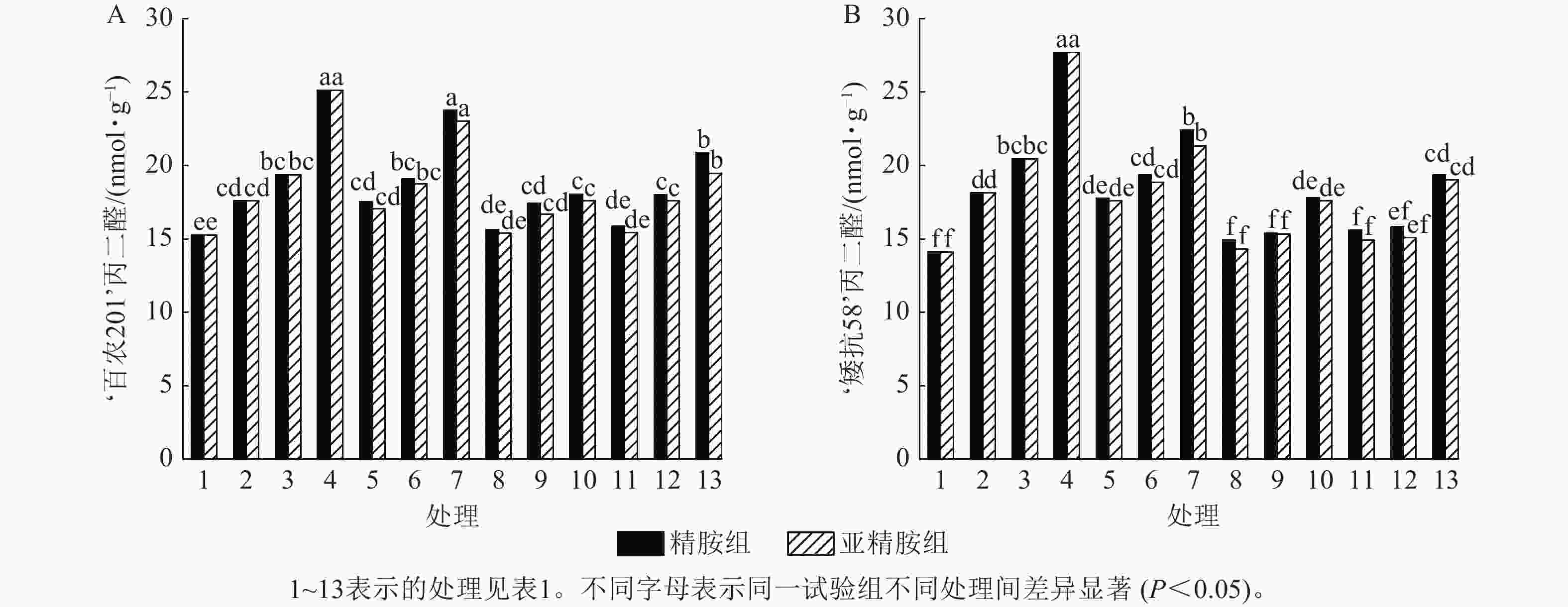
由图5可知:模拟干旱胁迫下,‘百农201’和‘矮抗58’的丙二醛质量摩尔浓度随着PEG-6000质量浓度的升高呈差异上升趋势。精胺浸种后,‘百农201’和‘矮抗58’的丙二醛质量摩尔浓度较对应干旱胁迫下的丙二醛质量摩尔浓度均有所下降。对‘百农201’,在0.10和0.20 mmol·L−1精胺可使质量浓度为15%PEG-6000干旱胁迫下的丙二醛质量摩尔浓度分别显著下降28.18%和16.92% (P<0.05);对‘矮抗58’,0.10和0.20 mmol·L−1精胺浸种对3种质量浓度干旱胁迫下的丙二醛产生均有显著抑制作用(P<0.05),其中在质量浓度为15%PEG-6000干旱胁迫下降幅最大,分别为35.80%和30.10%,即0.10 mmol·L−1精胺浸种效果优于0.20 mmol·L−1。亚精胺浸种结果与精胺趋势一致,0.10和0.20 mmol·L−1亚精胺浸种对3种质量浓度干旱胁迫下‘百农201’和‘矮抗58’的丙二醛产生均有显著缓解作用(P<0.05),其中对质量浓度为15%PEG-6000干旱胁迫下‘百农201’的丙二醛产生降幅分别为30.02%和22.53%,对‘矮抗58’的丙二醛产生降幅分别为36.56%和31.36%。以上结果表明:适宜浓度的精胺和亚精胺浸种可有效抑制干旱胁迫下丙二醛质量摩尔浓度的积累,其中0.10 mmol·L−1亚精胺对高质量浓度干旱胁迫下的缓解效果更佳。
-
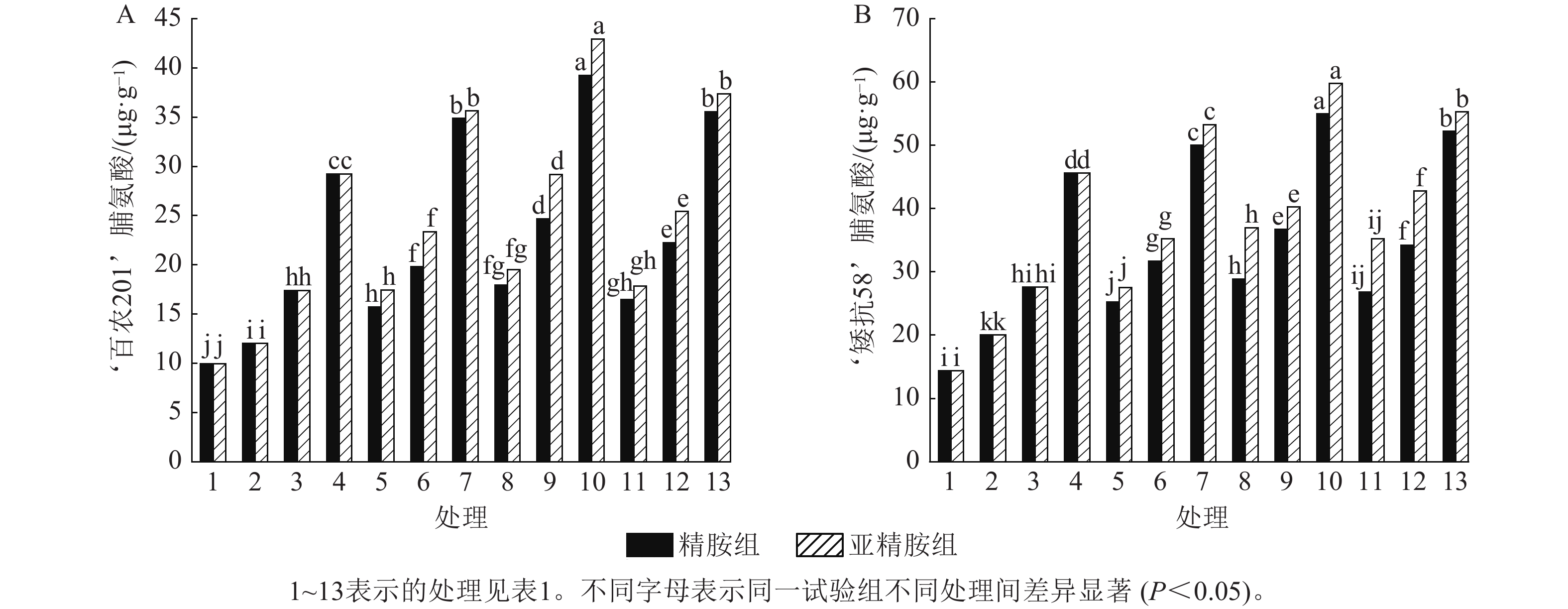
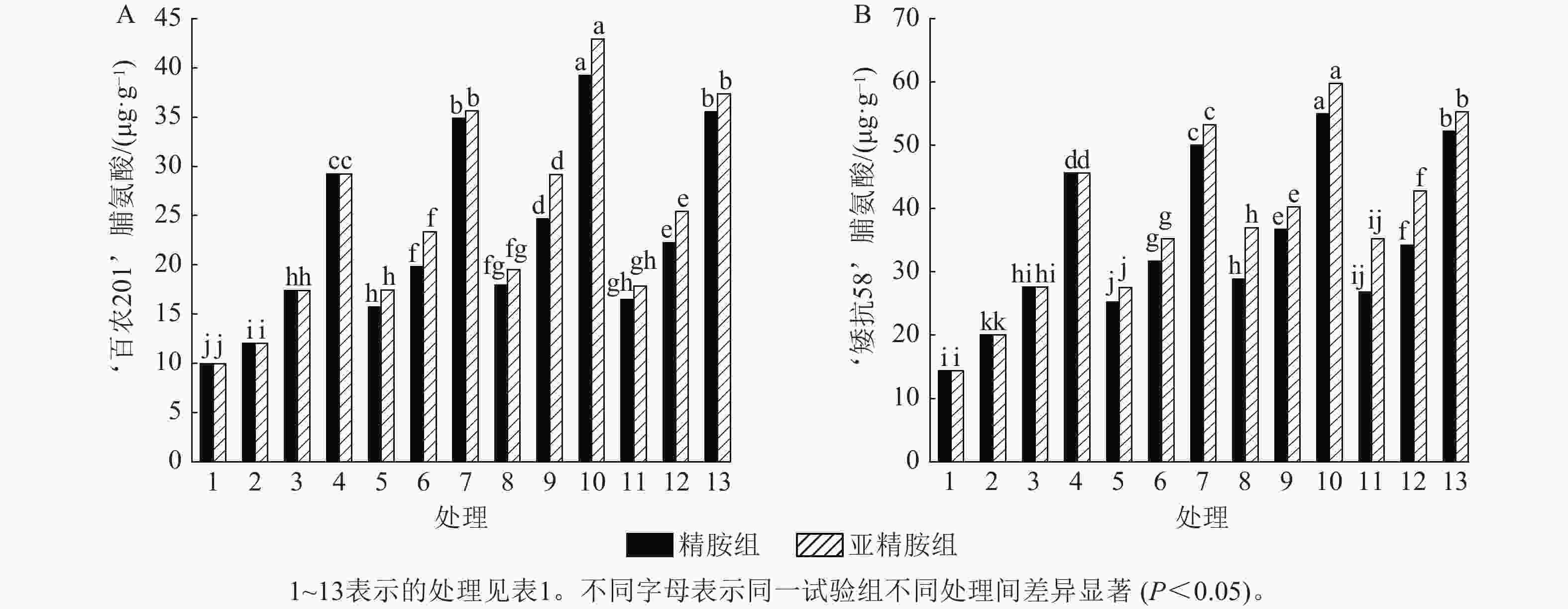
从图6可知:模拟干旱胁迫下,小麦脯氨酸质量分数随着PEG-6000质量浓度的升高呈上升趋势。在PEG-6000质量浓度为5%、10%和15%的模拟干旱胁迫下,‘百农201’脯氨酸质量分数较对照分别增加20.70%、74.77%和193.67%,‘矮抗58’分别增加39.42%、92.34%和217.48%,表明抗旱性较强的‘矮抗58’脯氨酸质量分数积累能力优于抗旱性较弱的‘百农201’。3种不同浓度的精胺和亚精胺浸种后,‘百农201’和‘矮抗58’脯氨酸质量分数均较对应质量浓度PEG-6000胁迫下的脯氨酸质量分数有显著增加(P<0.05),其中0.10 mmol·L−1精胺和亚精胺浸种对质量浓度为15%PEG-6000干旱胁迫下‘百农201’和‘矮抗58’的脯氨酸质量分数增幅最大,而且亚精胺的促进效果优于精胺。
-
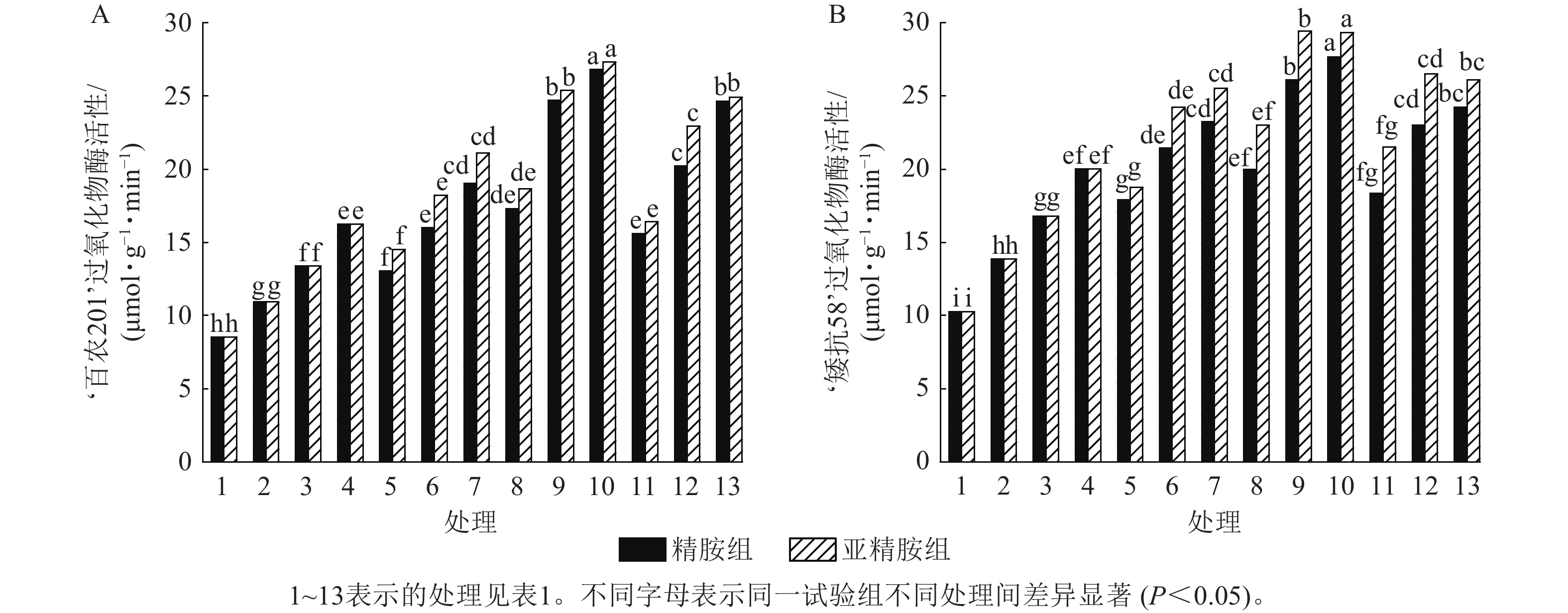
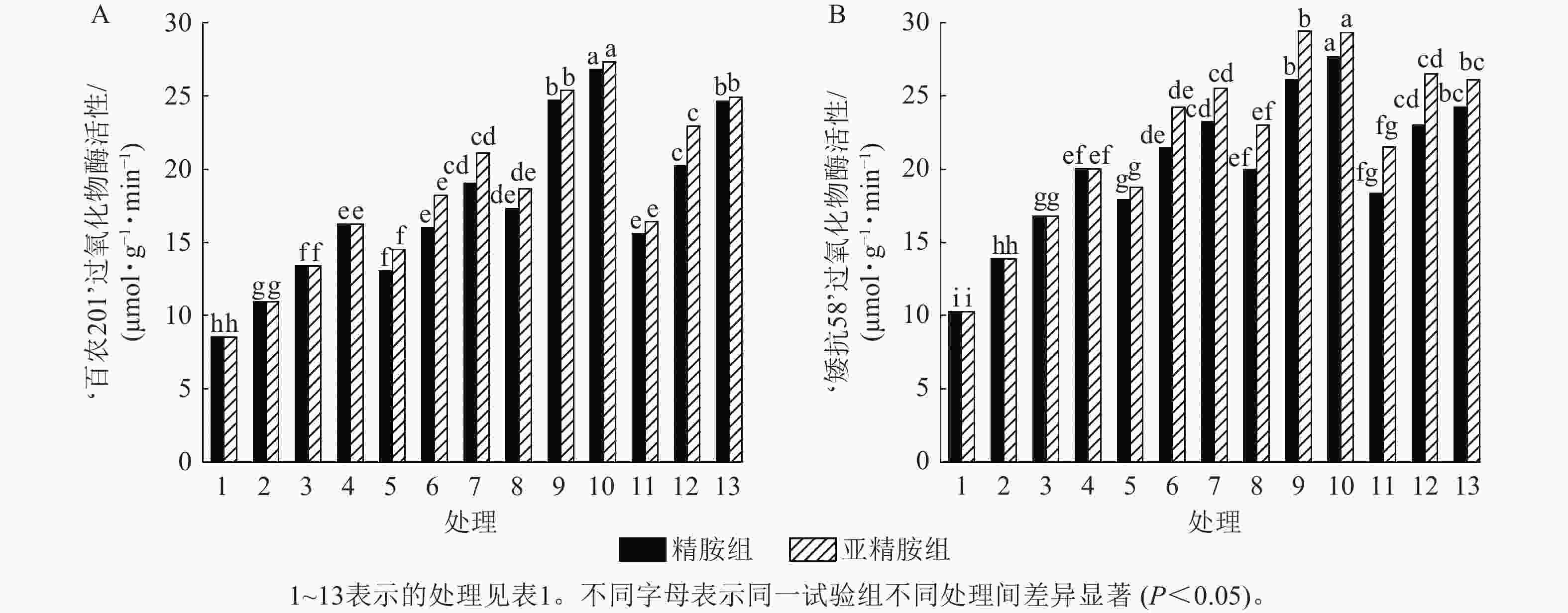
由图7可知:模拟干旱胁迫下,小麦过氧化物酶活性随着PEG-6000质量浓度的升高而呈上升趋势。在PEG-6000质量浓度为5%、10%和15%干旱胁迫下,‘百农201’过氧化物酶活性较对照增幅分别为27.87%、56.56%和90.28%,‘矮抗58’增幅分别为35.05%、63.49%和94.84%。3种不同浓度的精胺和亚精胺浸种后,‘百农201’和‘矮抗58’过氧化物酶活性较对应质量浓度PEG-6000胁迫下的过氧化物酶活性均有显著增加(P<0.05),其中0.10 mmol·L−1精胺和亚精胺浸种对高质量浓度干旱胁迫下‘百农201’和‘矮抗58’的过氧化物酶活性增幅最大,亚精胺浸种缓解效果较精胺好。
-
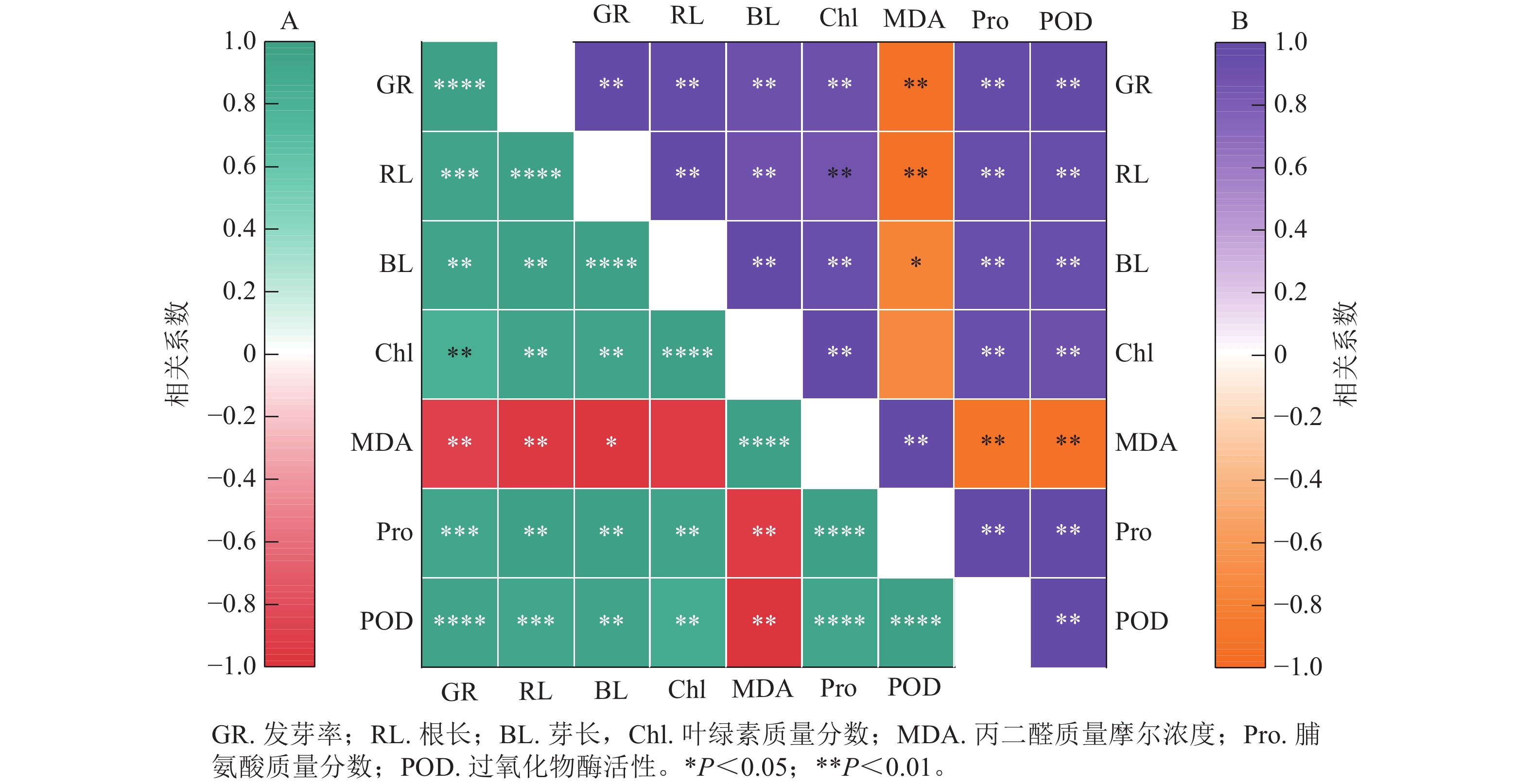
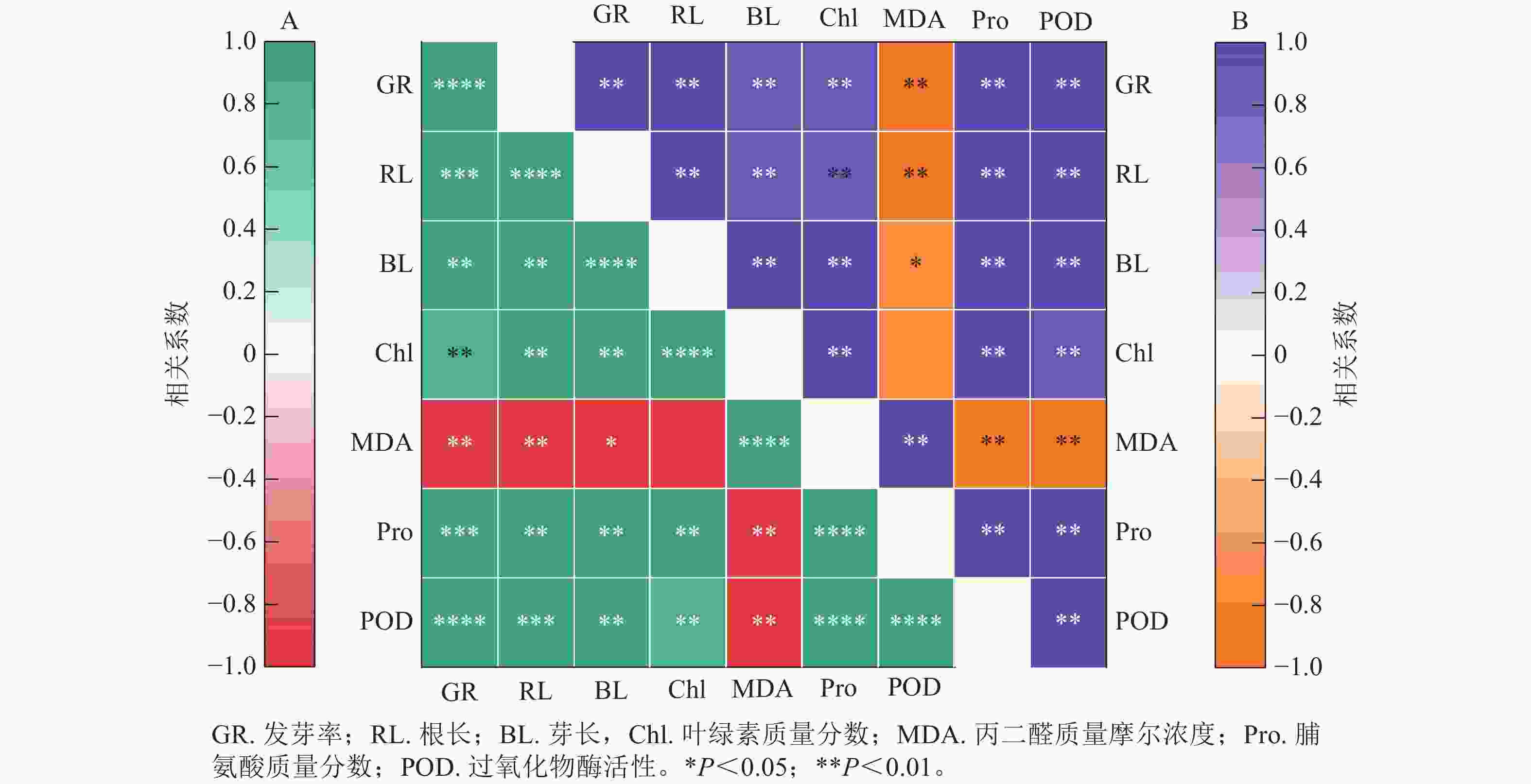
图8结果显示:精胺和亚精胺处理下小麦的发芽率与根长、芽长、脯氨酸质量分数和过氧化物酶活性均呈极显著正相关(P<0.01),与叶绿素质量分数呈显著正相关(P<0.05)。图8A表明:在精胺处理下,丙二醛质量摩尔浓度与发芽率、根长、芽长、叶绿素质量分数、脯氨酸质量分数、过氧化物酶活性均呈极显著负相关(P<0.01);图8B表明:在亚精胺处理下,丙二醛质量摩尔浓度与发芽率、根长、芽长、脯氨酸质量分数、过氧化物酶活性呈显著负相关(P<0.05),与叶绿素质量分数无显著相关。
-
由表2可知:不同浓度的精胺和亚精胺对小麦的干旱缓解均有显著提升,且在3种不同质量浓度PEG-6000干旱胁迫下,0.10 mmol·L−1浓度的亚精胺对‘百农201’和‘矮抗58’幼苗毒害的缓解作用均最显著。
PEG-6000
质量浓度/%精胺/
(mmol·L−1)亚精胺/
(mmol·L−1)‘百农201’ ‘矮抗58’ 隶属
函数值排名 隶属
函数值排名 5 0 0 0.058 4 7 0.086 3 7 0.05 0 0.281 7 6 0.343 2 6 0.10 0 0.721 7 3 0.746 4 2 0.20 0 0.435 1 5 0.380 6 5 0 0.05 0.583 5 4 0.502 4 4 0 0.10 0.987 9 1 0.998 2 1 0 0.20 0.746 3 2 0.596 6 3 10 0 0 0.020 7 7 0.039 8 7 0.05 0 0.316 6 6 0.283 5 6 0.10 0 0.794 7 2 0.721 3 3 0.20 0 0.429 1 5 0.431 3 5 0 0.05 0.451 9 4 0.470 5 4 0 0.10 0.981 2 1 1.000 0 1 0 0.20 0.596 6 3 0.724 3 2 15 0 0 0.028 4 7 0.047 2 7 0.05 0 0.323 9 6 0.319 8 6 0.10 0 0.840 7 2 0.734 2 2 0.20 0 0.496 4 4 0.406 9 5 0 0.05 0.463 7 5 0.501 0 4 0 0.10 0.997 3 1 1.000 0 1 0 0.20 0.629 7 3 0.625 4 3 Table 2. Membership function values of ‘Bainong 201’ and ‘Aikang 58’ under different treatments
-
种子萌发期与幼苗期是植物生长发育过程中对环境胁迫最为敏感的2个阶段,同时也是开展胁迫早期基础鉴定的关键时期[21−22]。有研究表明:干旱缺水会对小麦种子的萌发以及幼苗的生长产生抑制作用,而多胺作为种子引发剂,可有效缓解干旱胁迫对种子萌发造成的影响[4, 9, 23]。本研究中,PEG-6000模拟的干旱胁迫抑制了小麦种子的萌发、根长和芽长的生长,且随着PEG-6000质量浓度的增加而抑制作用显著增大,这与裴冬丽等[24]、GHOLIZADEH等[25]的研究结果一致,说明缺水引发的渗透胁迫会抑制植物生长。同时,本研究进一步证实了外源精胺和亚精胺浸种在一定程度上可以显著提高小麦在干旱胁迫下的发芽率、根长和芽长,这与外施精胺和亚精胺可缓解干旱胁迫对小麦[24−25]、玉米[26]、甜高粱Sorghum bicolor[27]和燕麦Avena sativa[28]等幼苗萌发生长的结果一致,说明适宜浓度的外源精胺和亚精胺浸种,可通过促进种子在短期内吸收、积累并转化上述物质,同时激活相关调控信号通路,从而有效促进干旱胁迫下种子的萌发与生长。因此,该处理可作为种子引发剂,用于抵御一定程度的干旱胁迫。此外,本研究中亚精胺浸种对‘百农201’在干旱胁迫下的缓解效果更明显,尤其0.10 mmol·L−1亚精胺效果最佳,说明亚精胺对抗旱性敏感品种的作用效果好于抗旱性强的品种,这与裴冬丽等[24]和张美微等[26]的研究结果一致,外源亚精胺能更好地缓解干旱胁迫对抗旱性弱的小麦和玉米品种生长的抑制作用,这很可能得益于亚精胺分子量相对较小、易于扩散,以及所带电荷较少、结合能力相对较弱等特点,使其在胁迫条件下能迅速发挥生理作用。
-
有研究表明:植物进行光合作用的能力与其叶绿素质量分数紧密相连,干旱胁迫往往会使与光合作用相关的膜结构受损,同时导致光合色素含量下降[15, 29],而多胺可提高植株在干旱胁迫下的光合色素含量,并维持叶绿体的超微结构[15]。在本研究中,‘百农201’和‘矮抗58’的叶绿素质量分数均随着PEG-6000质量浓度的增加而显著降低,表明干旱胁迫导致叶绿体损伤,叶绿素合成受阻,或相关叶绿素酶活性提高而使叶绿素进一步降解。本研究发现:外源精胺和亚精胺浸种可缓解干旱胁迫下小麦叶绿素质量分数的减少,这与众多研究结果相一致,如在胁迫条件下,添加外源多胺可提高紫花苜蓿Medicago sativa[30]、甜瓜Cucumis melo[31]、白三叶Trifolium repens[32]、烤烟Nicotiana tabacum[33]及玉米[34]等幼苗的叶绿素质量分数。这表明外源多胺能够通过调节叶绿体抗氧化系统,抑制干旱胁迫对叶绿体结构的破坏,有效减轻叶绿素合成受阻的程度,从而促进叶绿素合成。进一步比较发现:亚精胺浸种对干旱胁迫下叶绿素质量分数增加效果优于精胺,且亚精胺对抗旱性敏感品种‘百农201’的作用效果强于抗旱性较强的‘矮抗58’,这与李丽杰等[34]的研究结果一致,这种差异可能与品种的遗传特性有关。在干旱胁迫下,亚精胺的作用能够增强抗旱性较弱品种的自我调节能力,从而帮助其适应或缓解所遭受的胁迫。
-
植物在逆境中生长时,体内会积累大量丙二醛,其质量摩尔浓度与植株遭受胁迫强度及膜脂过氧化损伤程度均呈正相关[15]。脯氨酸作为抗氧化物质清除活性氧,在维持细胞膜系统的稳定中发挥着关键作用,其质量分数高低通常是鉴定植物抗逆性强弱的重要生理指标之一[15]。过氧化物酶是植物体内最重要的保护酶系统中的抗氧化酶之一,其活性水平直接反映了植物清除胁迫条件下积累的活性氧(ROS)能力。在逆境中,过氧化物酶通过高效清除活性氧,有效防止膜脂过氧化产生的膜系统损伤,从而更好地抵御外界胁迫、增强抗逆性。本研究表明:干旱胁迫下‘百农201’和‘矮抗58’幼苗叶片中丙二醛质量摩尔浓度、脯氨酸质量分数和过氧化物酶活性均较对照明显增加,且随着PEG-6000质量浓度的增加而显著上升,表明干旱胁迫导致膜脂过氧化作用,破坏了细胞膜结构,使活性氧的生成和清除失衡,同时通过积累大量的脯氨酸和提高过氧化物酶活性来维持较高的胁迫抗性,这一结果与杜红阳等[35]探究渗透胁迫下2种不同抗旱性小麦幼苗叶片脯氨酸质量分数变化规律时得到的结论一致,分析认为抗旱性较强的品种可能通过提高脯氨酸合成途径中的关键酶活性,从而促进脯氨酸的进一步积累,以此维持细胞渗透平衡。结合生长指标在干旱胁迫下受抑制的表现可知:小麦幼苗通过提高自身抗氧化酶活性,仍不足以清除过多的活性氧,从而无法避免其带来的伤害。精胺和亚精胺具有中和酸、抗氧化及稳定细胞膜的能力,本研究利用3种不同浓度的外源精胺和亚精胺进行浸种处理,发现‘百农201’和‘矮抗58’丙二醛质量摩尔浓度较对应干旱胁迫下的丙二醛质量摩尔浓度有所下降,脯氨酸质量分数和过氧化物酶活性均进一步显著提高,其中0.10 mmol·L−1的精胺和亚精胺浸种涨幅最大,且亚精胺浸种比精胺浸种在干旱胁迫下产生的缓解效果更佳。这与干旱胁迫下或逆境胁迫中多胺对紫花苜蓿[30]、甜瓜[31, 36]、白三叶[32]、烤烟[33]、甜高粱[27]、燕麦[28]、小麦[37]、南瓜Cucurbita moschata[38]、假俭草Eremochloa ophiuroides[39]、赤霞珠Vitis vinifera[40]、蚕豆Vicia faba[41]及红花Carthamus tinctorius[14]等幼苗的结果一致。表明适宜浓度的精胺和亚精胺浸种能够有效降低植物在干旱胁迫中活性氧的积累,抑制小麦幼苗细胞膜质的氧化,从而减少了丙二醛的进一步产生;同时通过提高植物体内合成脯氨酸的关键酶活性,促进脯氨酸质量分数进一步积累,减轻干旱胁迫对膜结构和功能的损伤或毒害作用;通过提高植物体内抗氧化系统活性,清除体内积累的过量活性氧,有效减轻膜脂过氧化损伤,增强植株萌发期的抗旱性。
-
相关性分析显示:精胺和亚精胺浸种处理下小麦的发芽率与根长、芽长、脯氨酸质量分数和过氧化物酶活性均呈极显著正相关,与叶绿素质量分数呈显著正相关。LI等[42]研究发现:亚精胺可通过减轻氧化损伤、维持能量稳态调控小麦抗逆性,这与本研究中过氧化物酶活性提升、丙二醛质量摩尔浓度降低的规律相呼应;GHOLIZADEH等[25]发现:外源亚精胺能显著提高小麦种子发芽率,促进脯氨酸积累并增强 过氧化物酶活性,这与本研究中发芽率分别与脯氨酸质量分数、过氧化物酶活性呈极显著正相关的结果相呼应;HASSAN等[37]研究表明:外源多胺通过保护细胞膜与叶绿体超微结构缓解干旱,进一步明确了多胺调控下小麦生长指标与生理指标相关性的内在生理机制。
-
综上所述,PEG-6000模拟的干旱胁迫抑制小麦幼苗的生长发育,使发芽率、根长、芽长、叶绿素质量分数下降,丙二醛质量摩尔浓度、脯氨酸质量分数和过氧化物酶活性升高。适宜浓度的精胺和亚精胺浸种可促进干旱胁迫下种子的发芽、根长和芽长的生长以及叶绿素质量分数的提高,同时使丙二醛质量摩尔浓度下降,脯氨酸质量分数和过氧化物酶活性进一步升高,从而稳定细胞内环境,缓解干旱胁迫产生的生长抑制作用。精胺和亚精胺浸种对缓解干旱胁迫下抗旱性较弱的‘百农201’作用效果好于抗旱性较强的‘矮抗58’,相较于精胺,亚精胺能更好地减轻干旱对幼苗的胁迫作用。综合分析表明:0.10 mmol·L−1亚精胺对干旱胁迫下幼苗毒害的调节效应最显著。
Effects of exogenous spermine and spermidine seed soaking on wheat germination and growth under drought stress
doi: 10.11833/j.issn.2095-0756.20250337
- Received Date: 2025-06-13
- Accepted Date: 2026-03-23
- Rev Recd Date: 2025-07-18
-
Key words:
- wheat /
- drought stress /
- exogenous spermine /
- spermidine /
- physiological characteristics
Abstract:
| Citation: | YANG Xiaofei, ZHANG Mengyan, ZHANG Shujun. Effects of exogenous spermine and spermidine seed soaking on wheat germination and growth under drought stress[J]. Journal of Zhejiang A&F University, 2026, 43(X): 1−12 doi: 10.11833/j.issn.2095-0756.20250337 |









 DownLoad:
DownLoad: