-
露地蔬菜是中国蔬菜种植的主要类型,种植面积占全国蔬菜总种植面积的85%[1]。然而,随着栽培年限延长和栽培面积扩大,高强度种植模式易使土壤发生次生盐渍化、养分失衡和土传病害等问题,导致土壤微生态环境发生改变[2],已成为制约露地农业可持续发展的重要因素。露地蔬菜地大量施用化肥后,土壤在短期内呈现出有效养分的迅速提高,但长期高强度种植会导致土壤酸化加剧、有机碳库消耗加速、有益微生物群落衰退以及土传病原菌积累等问题,威胁作物健康。单纯依赖土壤自身难以抵御这些长期退化风险,因此,土壤调理剂与有机肥成为破解上述问题的具有针对性的关键技术手段。
土壤调理剂通过其独特的物理和化学特性调节土壤理化性质,优化土壤微生物群落结构,其作用速度快、施用便捷且成本较低,被广泛用于健康土壤培育和土传病害防治[3]。目前,调理剂主要有无机和有机两大类。无机调理剂以黏土矿物和石灰类为主;有机调理剂包括生物炭和有机物料等类型[4]。石灰类无机调理剂虽能快速提高土壤pH,但由于其在土壤中迁移性较差,长期大量施用易导致土壤板结、营养元素比例失调,进而导致作物产量下降[5]。牡蛎Crassostrea gigas壳等天然含钙调理剂不仅碳酸钙质量分数高达90%以上,还含有铁、铜、锌等多种微量元素[6]。经高温焙烧处理后,牡蛎壳中碳酸钙转化成氧化钙,形成复杂的多孔结构,比表面积随之增大,因此具备较强的吸附和交换能力,可钝化重金属,对土壤具有优良的改良作用[7]。已有研究表明:增施土壤调理剂可缓解土壤酸化,提高土壤pH与有机质,进而改善作物品质[8]。
有机肥中富含植物生长所需的有益物质和营养元素,不仅可以抑制土壤酸化,提升土壤养分水平,为作物生长提供养分支持,还能增加土壤有机质,优化有机质组成,改善土壤理化性质与微生物活性。因此,施用有机肥被视作土壤培肥,推动农业循环发展及维护土壤健康的重要措施[9]。相关研究表明:增施有机肥能降低土壤pH,显著提高土壤中速效养分及有机质,促进作物生长发育,改善土壤微生态环境,并提高蔬菜产量和品质[10]。
土壤微生物在改善土壤结构,调节土壤养分平衡,维持土壤生态平衡和影响作物生产等方面发挥着关键作用[11],同时也是衡量土壤质量的重要指标[12]。施用调理剂不仅能够通过调节土壤理化性质为微生物提供营养来源,促进微生物生长繁殖,还能改善土壤环境、提高微生物活性与多样性,进而优化微生物群落结构。有机肥中富含丰富的碳、氮和微量元素,为土壤微生物生长所必需,并能优化土壤微生物群落结构和功能[13]。有研究表明:调理剂中的钙、镁等元素可增强植物的抗病性,同时抑制镰刀菌属 Fusarium、丝核菌属 Rhizoctonia等病原真菌,并为芽孢杆菌属 Bacillus、假单胞菌属 Pesudomonas等有益微生物提供碳源。CHEN等[14]研究发现:有机肥与调理剂配施能显著提高细菌多样性,增加土壤中放线菌门 Actinobacteriota和变形菌门 Proteobacteria相对丰度。LI等[15]研究发现:有机肥配施调理剂能够增加土壤有机碳,促进有益菌——芽孢杆菌的增殖。然而,关于如何合理施用土壤调理剂和有机肥,以及两者配施对露地蔬菜土壤养分及微生物群落的影响,仍有待深入研究。
本研究在田间条件下探究不同施用量土壤调理剂与有机肥配施对露地蔬菜土壤养分的影响,同时运用高通量测序技术分析不同处理下露地蔬菜土壤细菌的多样性、群落结构及生态互作网络特征的变化规律,揭示驱动这些变化的主要环境因素,为今后露地蔬菜高效培肥和健康可持续发展提供理论依据。
-
试验地位于浙江省杭州市临安区绿源蔬菜基地(30°08′21 " N,119°05′20 " E),试验于2023年5月开始,种植蔬菜为茄Solanum melongena。本研究试验地长期露地种植蔬菜,由于长期大量施用化肥,土壤质量明显下降。试验采用双因素随机区组设计,主因素为调理剂施用梯度,副因素为有机肥施用梯度。设置3个调理剂施用梯度:不施用调理剂(NSC)、低调理剂处理(0.75 t·hm−2,LSC)、高调理剂处理(1.50 t·hm−2,HSC)。根据试验地基础肥力及浙江省耕地土壤健康评价指标体系的推荐用量,在每个调理剂施用梯度下分别设置3个有机肥施用量处理:不施用有机肥(NOF)、低有机肥处理(4.5 t·hm−2,LOF)、高有机肥处理7.5 (t·hm−2,HOF)。土壤调理剂选用特贝钙调理剂,以牡蛎壳为原料,为无机调理剂,购自福建省玛塔农业发展有限责任公司,氧化钙(CaO)质量分数≥45.0%、碳酸钙(CaCO3)质量分数为40.0%~45.0%,总钙质量分数为38.5%~40.2%。有机肥购自临安锦来富农业有限公司,有机质质量分数≥45.0%,氮(N)质量分数+五氧化二磷(P2O5)质量分数+氧化钾(K2O)质量分数≥5.0% (P2O5为1.8%~2.0%),含固氮菌、解磷菌及螯合态中微量元素。高有机肥处理虽带入一定量磷,但并未提高土壤中有效磷,反而使其降低,可能是由于该有机肥为山核桃蒲壳发酵而成,碱性较强,导致土壤磷有效性降低。同时,山核桃蒲壳的吸附性较强,也是导致土壤有效磷降低的可能原因之一。各小区面积均为32 m2,每个处理3个重复,共9个处理,27个小区。起垄覆膜露天种植茄,其中水分管理采用自然降水+人工沟灌协同调控,定植前7 d将氮肥与调理剂、有机肥混合沟施,基肥施用时将调理剂、有机肥撒施后旋耕;在生育期进行双干整枝,植株绑缚防倒伏。除调理剂和有机肥施用处理外,所有小区采用一致的常规栽培管理。
试验为期3个月,在茄成熟采收后进行采样,且在试验地茄收获后,秸秆未还田。每个试验小区用土钻对角线五点取样法采集0~20 cm耕层土样,去除杂质均匀混合置于冰盒内带回实验室,过2 mm筛后分装,一份于−80 ℃超低温冰箱保存,用于提取DNA并测定土壤细菌群落结构;另一份风干,用于测定土壤理化性质。
-
参考鲁如坤《土壤农业化学分析方法》[16]测定土壤理化性质。采用玻璃电极法测定土壤pH,土水比为1.0∶2.5;采用乙酸铵浸提-火焰光度计测定土壤速效钾(AK);采用碳酸氢钠浸提,钼锑抗比色法测定土壤有效磷(AP);采用碱解扩散法测定土壤碱解氮(AN);用浓硫酸-重铬酸钾外加热法测定土壤有机质(SOM);采用凯氏定氮法测定土壤全氮(TN);采用三氯化六氨合钴浸提,分光光度法测定土壤阳离子交换量(CEC)。
-
采用MoBio PowerSoil® DNA Isolation Kit试剂盒(MoBio Laboratories)提取土壤总DNA,称取0.5 g冻干土样,根据试剂盒步骤进行DNA提取。对提取后的DNA通过微量分光光度计(NanoDrop ND-1000)测定其浓度,将其存放于−40 ℃冰箱保存,为后续分析备用。委托杭州联川生物技术股份有限公司进行土壤DNA样品的高通量测序。使用引物338F (5′-ACTCCTACGGGAGGCAGCAG-3′)和806R (5′-GGACTACHVGGGTWTCTAAT-3′)通过聚合酶链式反应(PCR)对16S rDNA 基因的V3~V4可变区进行扩增,再基于Illumina NovaSeq平台对测序样本实施双端测序。序列处理阶段借助QIIME2 软件中的DADA2插件对所有原始序列(input)依次进行质量控制(filtered)、去噪(denoised)、拼接(merged)及去嵌合体(non-chimeric)处理,最终形成扩增子序列变体(ASV),物种注释借助Silva 数据库(16S)完成,且将置信度阈值设定为0.7。当各样品的Alpha多样性稀释曲线(rarefaction curve)趋于平缓,表明该样品所含的绝大部分的微生物多样性信息已被充分反映,此时测序数据量已达到充分程度。
-
采用SPSS 26.0进行数据的双因素方差分析(two-way ANOVA),并通过邓肯法(Duncan)检验不同调理剂和有机肥施用量下土壤养分和微生物群落差异的显著性(P<0.05)。利用R 4.4.2 软件对微生物测序数据进行分析。基于细菌操作分类单元(OTU)丰度表,通过QIIME中alpha_diversity.py 脚本计算土壤细菌群落的Shannon指数和Chao 1指数。采用皮尔逊(Pearson)相关性分析探究土壤环境因子与土壤细菌群落门水平优势菌群之间的相关性。通过非度量多维标度分析(NMDS)解析土壤细菌群落结构组成特征。利用R语言vegan包进行冗余分析(RDA),探究不同施肥或调理剂施用量对细菌群落结构具有显著影响的环境因子;同时采用置换多元方差分析法(permutational multivariate analysis of variance,PERMANOVA)检验不同有机肥或调理剂处理间土壤细菌群落结构差异的显著性[17]。基于R语言的igraph包构建无向的相关性网络,借助Gephi 0.9.2绘制微生物网络图,同时计算相关网络拓扑指数,分析不同有机肥或调理剂处理下土壤细菌网络拓扑特征差异。
-
随土壤调理剂施用量的增加,土壤pH以及速效钾、有效磷、有机质质量分数均显著增加(P<0.05),但对土壤碱解氮、全氮质量分数以及阳离子交换量没有显著影响(表1)。随着有机肥施用量的增加,土壤有机质质量分数均显著增加,但有效磷显著下降(P<0.05),有机肥施用量对土壤速效钾、碱解氮、全氮质量分数及阳离子交换量、pH均没有显著影响。土壤调理剂和有机肥的施用对土壤速效钾、有效磷以及有机质质量分数具有显著的交互作用(P<0.05)。
处理 pH 速效钾/
(mg·kg−1)有效磷/
(mg·kg−1)碱解氮/
(mg·kg−1)有机质/
(g·kg−1)全氮/
(g·kg−1)阳离子效换量/
(cmol·g−1)NSC NOF 6.80±0.03 Cb 376.00±20.21 Ba 30.21±2.26 Aa 135.35±15.04 Aa 32.08±1.97 ABab 1.79±0.19 Aa 6.19±0.70 Aa LOF 6.75±0.01 Ab 417.00±10.39 Aa 16.10±1.77 Ab 115.30±5.01 Aa 29.31±0.41 Cb 1.54±0.25 Aa 5.54±1.05 Aa HOF 7.07±0.08 Aa 426.67±44.74 Aa 13.83±1.48 Bb 110.28±5.01 Aa 34.55±1.05 Ba 1.67±0.06 Aa 5.43±0.03 Ba LSC NOF 7.09±0.05 Ba 336.33±11.79 Bb 21.76±0.41 Ba 105.27±8.68 Aa 28.94±0.22 Bb 1.63±0.09 Aa 6.33±0.12 Aa LOF 6.87±0.12 Aa 513.00±11.93 Aa 22.00±2.70 Aa 125.32±5.01 Aa 36.95±0.42 Aa 1.77±0.05 Aa 5.93±0.49 Aa HOF 6.89±0.15 Aa 494.33±47.01 Aa 21.53±2.69 Ba 115.30±5.01 Aa 38.92±1.14 Aa 1.73±0.10 Aa 5.68±0.41 Ba HSC NOF 7.42±0.11 Aa 737.00±17.90 Aa 36.24±2.50 Aa 130.34±13.26 Aa 34.98±1.83 Aa 1.83±0.12 Aa 6.35±1.28 Aa LOF 7.12±0.14 Aa 455.00±47.92 Ab 20.59±4.59 Aa 110.28±5.01 Aa 32.19±0.88 Ba 1.75±0.12 Aa 6.38±0.44 Aa HOF 7.21±0.12 Aa 539.33±39.57 Ab 33.93±2.63 Aa 125.32±10.03 Aa 36.42±0.29 ABa 1.70±0.04 Aa 7.56±0.37 Aa SC *** *** *** ns ** ns ns OF ns ns *** ns *** ns ns SC×OF ns *** ** ns *** ns ns 说明:数据为平均值±标准差。不同大写字母表示在相同有机肥施用量条件下,不同调理剂施用量处理间差异显著(P<0.05);不同小写字母表示在相同土壤调理剂施用量条件下,不同有机肥施用量处理间差异显著(P<0.05)。*、**、***分别表示在0.05、0.01和0.001水平上差异显著;ns表示显著不差异。NSC. 不施调理剂;LSC. 低调理剂处理; HSC. 高调理剂处理。NOF. 不施有机肥;LOF. 低有机肥处理;HOF. 高有机肥处理。SC.有机肥处理;OF. 有机肥处理。 Table 1. Soil chemical properties and nutrient contents under different treatments
-
各处理组对土壤细菌群落Shannon指数和Chao 1指数无显著影响。双因素方差分析表明:施用有机肥显著提高了土壤细菌群落Chao 1指数(P<0.05),对土壤细菌Shannon指数无显著影响;施用调理剂对土壤细菌群落Shannon指数和Chao 1指数均无显著影响。调理剂和有机肥施用量对土壤细菌群落α多样性指数无显著交互作用(表2)。
处理 Shannon指数 Chao 1指数 处理 Shannon指数 Chao 1指数 NSC NOF 10.47±0.34 Aa 3446.10 ±702.30 AaHSC NOF 10.71±0.17 Aa 3709.75 ±545.85 AaLOF 11.04±0.10 Aa 4808.17 ±159.99 AaLOF 10.87±0.11 Aa 4531.79 ±191.07 AaHOF 10.97±0.08 Aa 4828.16 ±173.94 AaHOF 10.87±0.05 Aa 4325.87 ±269.98 AaLSC NOF 10.91±0.19 Aa 4200.94 ±642.55 AaSC ns ns LOF 10.97±0.04 Aa 4655.27 ±99.18 AaOF ns * HOF 10.90±0.08 Aa 4618.17 ±89.10 AaSC×OF ns ns 说明:数据为平均值±标准差。不同大写字母表示在相同有机肥施用量条件下,不同调理剂施用量处理间差异显著(P<0.05);不同小写字母表示在相同土壤调理剂施用量条件下,不同有机肥施用量处理间差异显著(P<0.05)。*、**、***分别表示在0.05、0.01和0.001水平上差异显著;ns表示显著不差异。NSC. 不施调理剂;LSC. 低调理剂处理; HSC. 高调理剂处理。NOF. 不施有机肥;LOF. 低有机肥处理;HOF. 高有机肥处理。SC.有机肥处理;OF. 有机肥处理。 Table 2. Diversity index of soil bacterial communities under different treatments
-
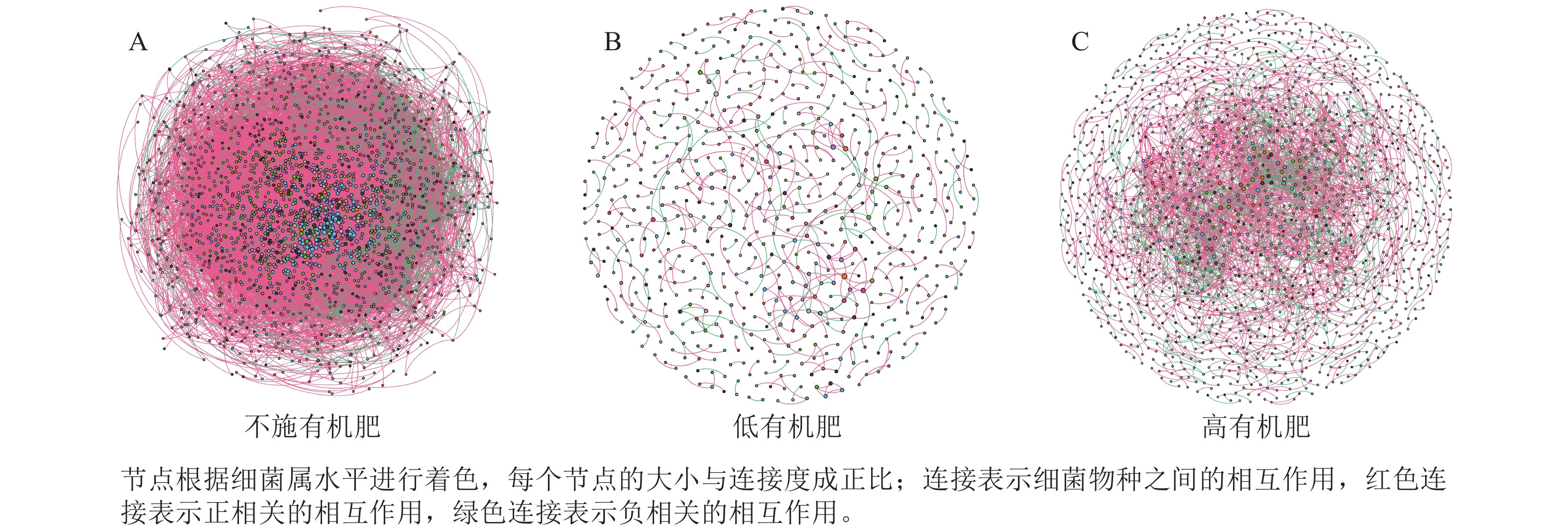
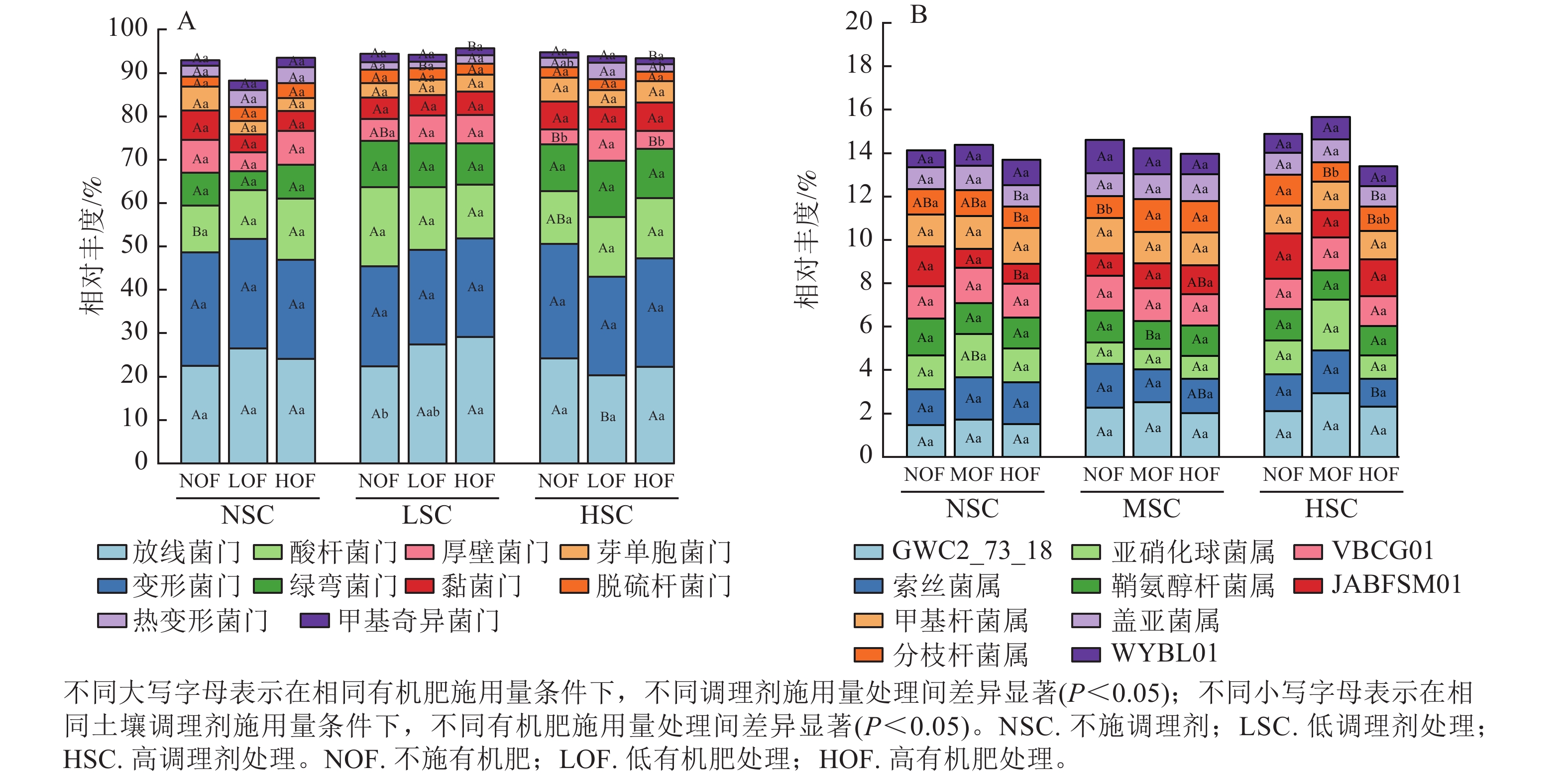
选取各处理组土壤细菌在门、属水平上相对丰度前10位的类群进行分析。从门水平来看,相对丰度前10位的类群包含放线菌门、变形菌门、酸杆菌门Acidobacteriota、绿弯菌门Chloroflexota、厚壁菌门Firmicutes、黏菌门Myxococcota、芽单胞菌门Gemmatimonadota和脱硫杆菌门Desulfobacterota、热变形菌门Thermoproteota和甲基奇异菌门Methylomirabilota等,累计相对丰度达到群落相对丰度的85%以上(图1A)。放线菌门和变形菌门为优势菌群,相对丰度分别为20.27%~29.13%和21.80%~26.40%。在不同施肥处理下,放线菌门、酸杆菌门、厚壁菌门、热变形菌门和甲基奇异菌门的相对丰度均存在显著差异(P<0.05)。在高调理剂处理下,低有机肥处理的土壤厚壁菌门和热变形菌门相对丰度显著高于其他处理(P<0.05)。
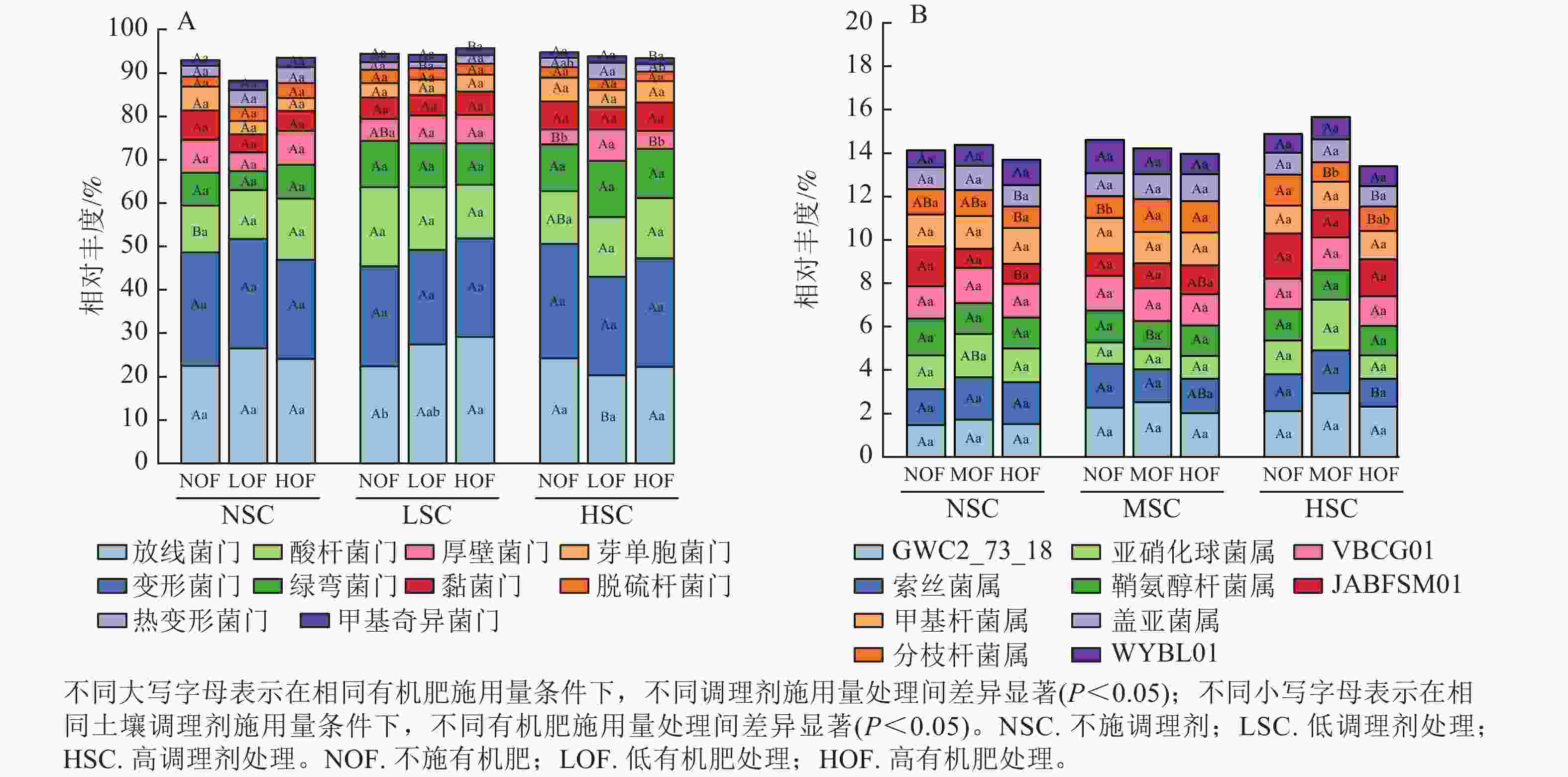
Figure 1. Community composition of soil bacteria at the phylum(A) and genus(B) level under different treatments
由图1B可知:不同施肥处理下优势菌属为GWC2_73_18、索丝菌属Kouleothrix、亚硝化球菌属Nitrosocosmicus、鞘氨醇杆菌属Sphingomicrobium、VBCG01、JABFSM01、甲基杆菌属Methyloceanibacter、分枝杆菌属Mycobacterium、盖亚菌属Gaiella和WYBL01等,其中以GWC2_73_18相对丰度最高,为1.48%~2.94%。其中索丝菌属、亚硝化球菌属、JABFSM01、分枝杆菌属和盖亚菌属在不同施肥处理下均有显著差异。在不施有机肥处理下,高调理剂处理显著提高了分枝杆菌属的相对丰度(P<0.05)。
-
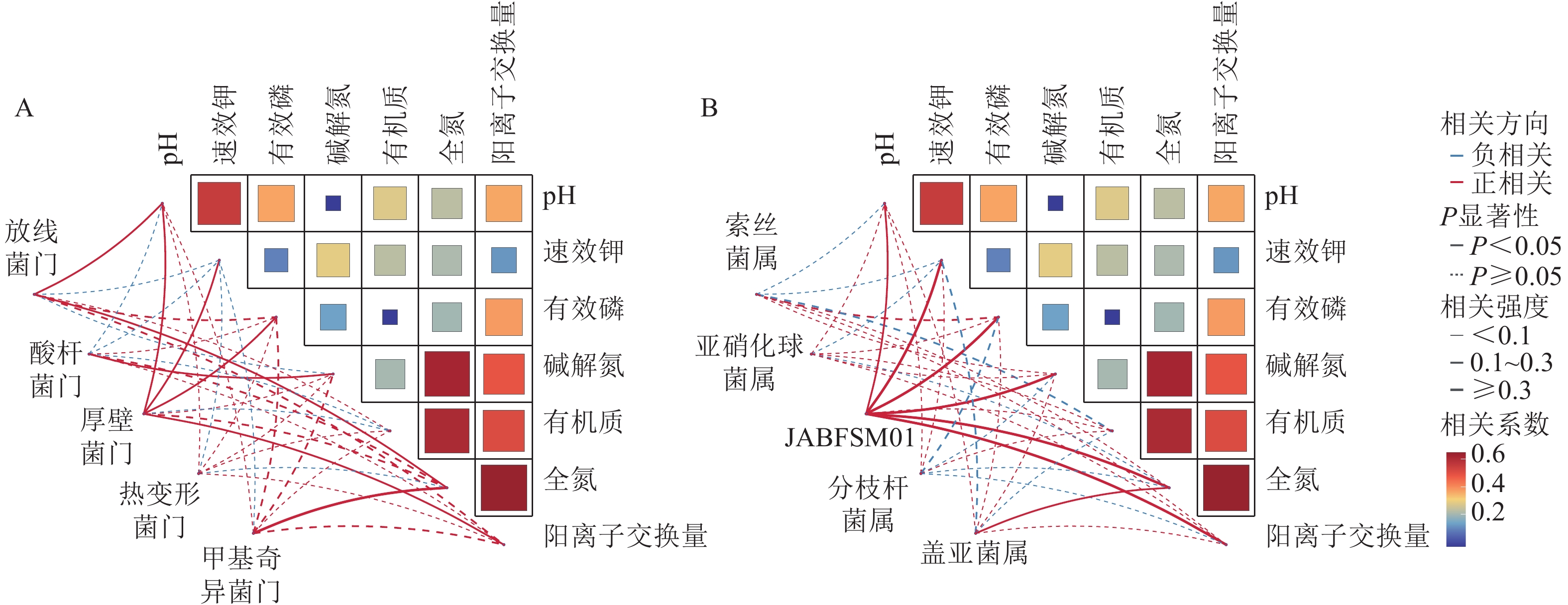
采用Pearson相关性分析法探究土壤环境因子与土壤细菌群落门水平优势菌群之间的相关性(图2A)。结果表明:土壤pH和速效钾质量分数与厚壁菌门的相对丰度呈显著正相关(P<0.05),与酸杆菌门的相对丰度呈显著负相关(P<0.05)。土壤有机质质量分数与放线菌门和厚壁菌门的相对丰度呈显著负相关(P<0.05)。
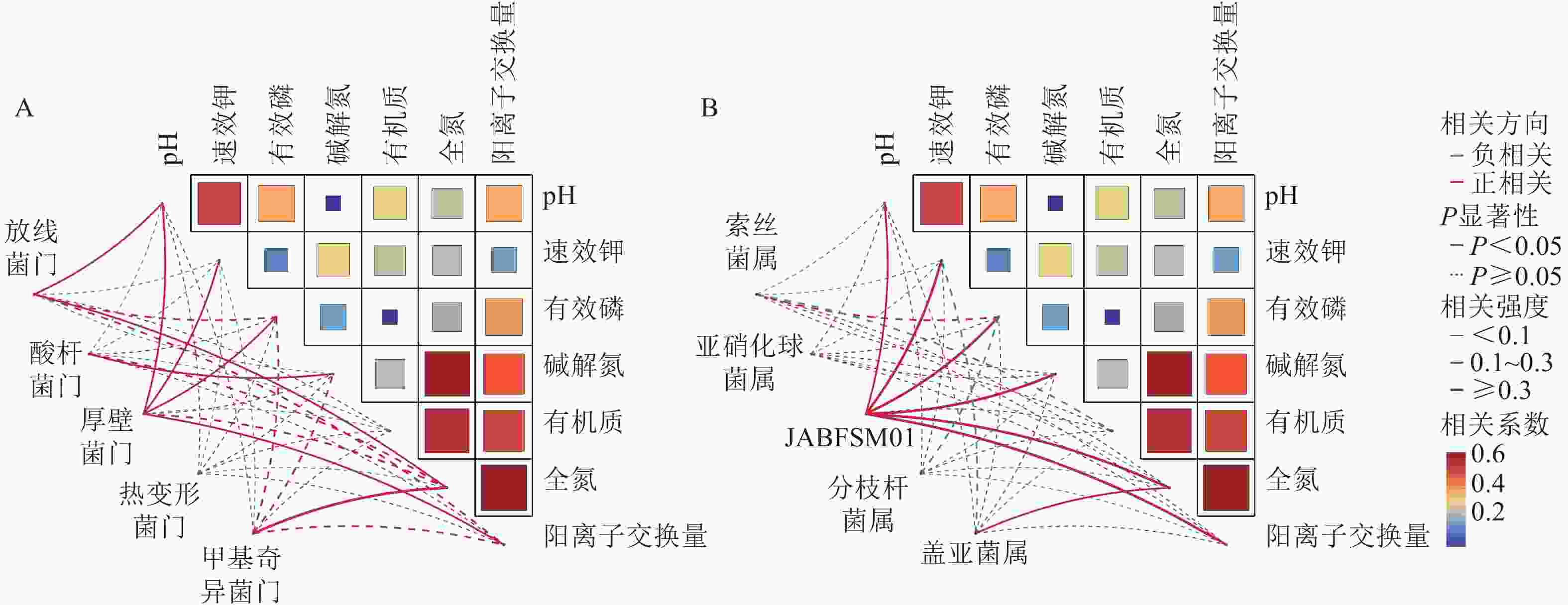
Figure 2. Correlation analysis of soil physicochemical properties with the dominant bacterial phyla(A) and genera(B)
由图2B可知,土壤全氮质量分数与JABFSM01和盖亚菌属的相对丰度呈显著正相关(P<0.05),与亚硝化球菌属和分枝杆菌属的相对丰度呈显著负相关(P<0.05)。土壤有机质质量分数与索丝菌属和盖亚菌属的相对丰度呈显著负相关(P<0.05)。
-
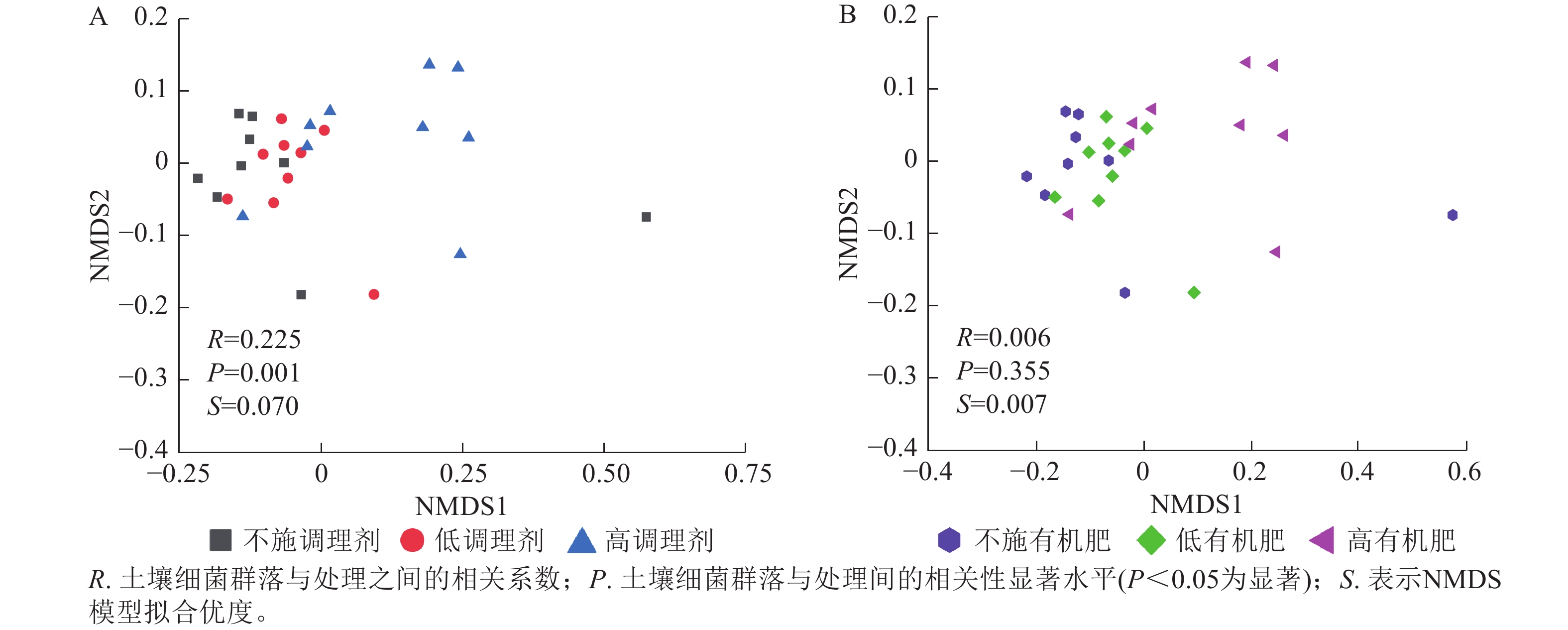
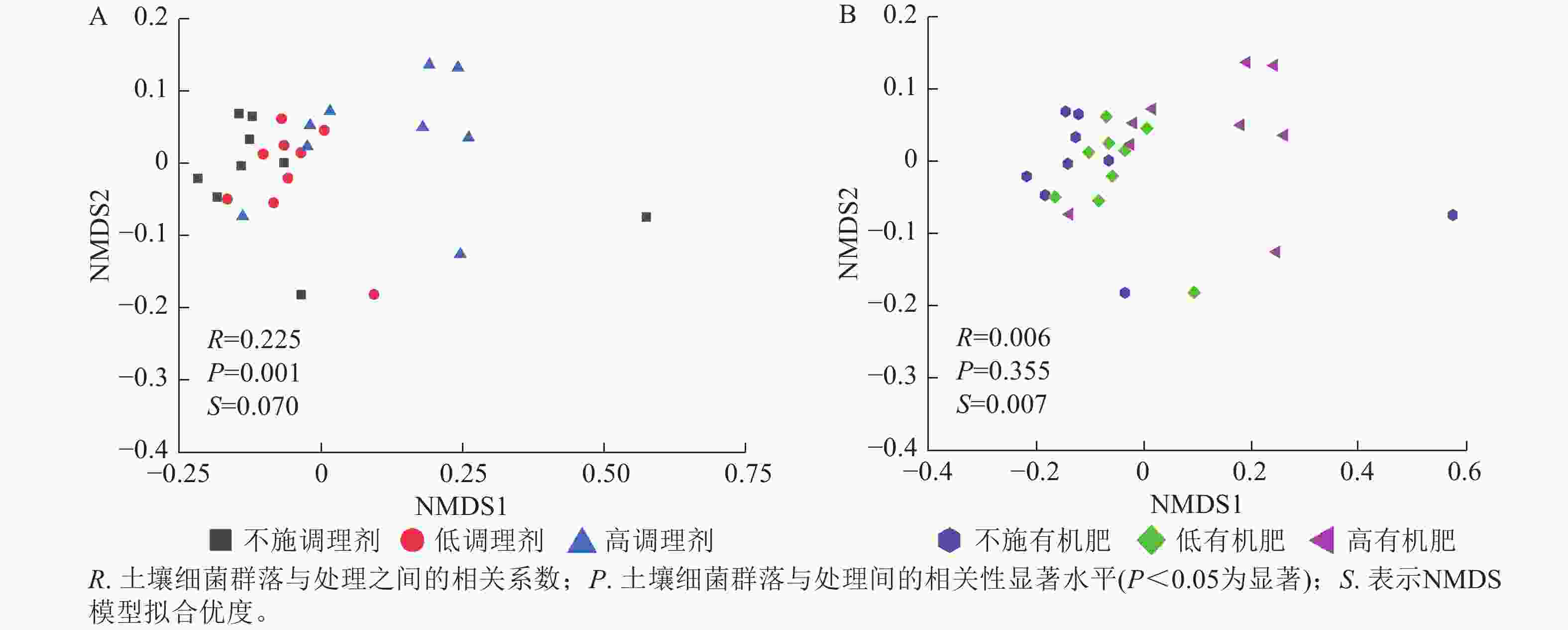
基于Bray-Curtis距离的土壤细菌群落非度量多维标度分析结果表明:不同调理剂施用量对细菌群落结构具有显著影响(P< 0.05),其中不施调理剂(R=0.11,P=0.007)和低调理剂处理(R=0.24,P=0.011)与高调理剂之间存在显著差异,而不施调剂处理与低调剂处理之间差异不显著(图3A)。不同有机肥施用量对土壤细菌群落结构没有显著影响(图3B)。
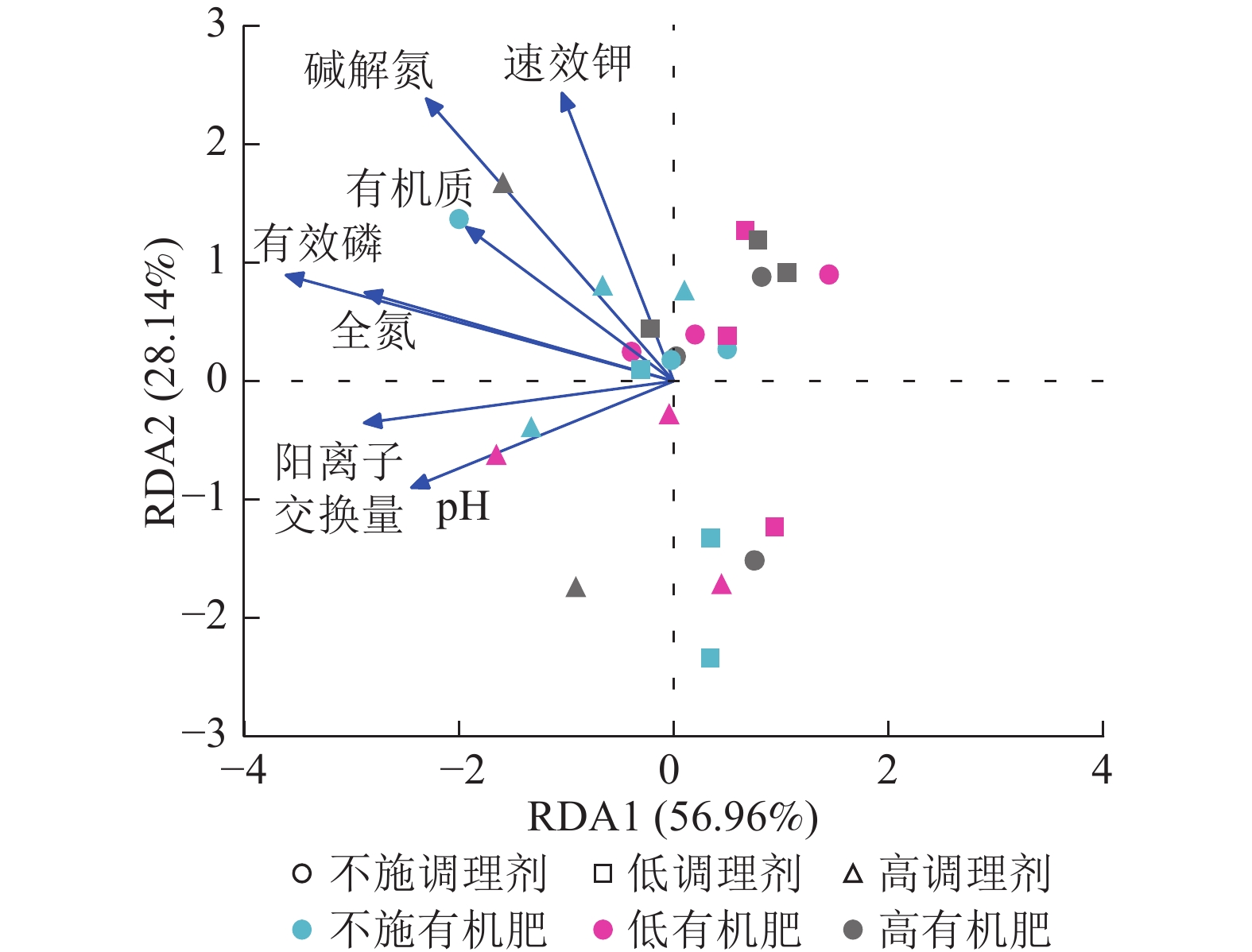
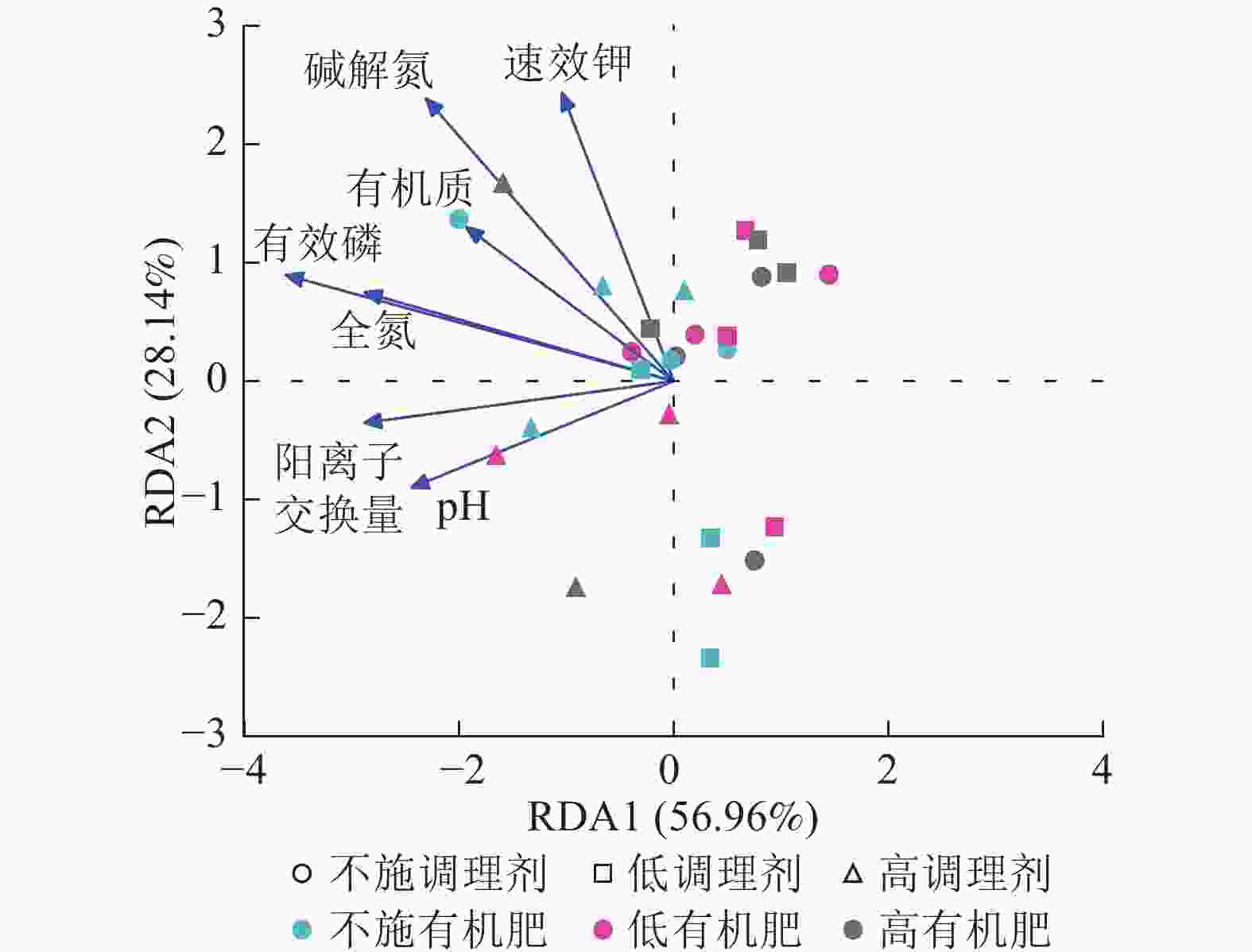
针对不同施肥模式下影响土壤细菌群落结构的环境因子进行冗余分析(图4)。结果表明:不同调理剂及有机肥施用量处理下,环境因子共解释了细菌群落结构变异的85.10%,轴1解释量为56.96%,轴2解释量为28.14%。其中土壤有效磷质量分数与土壤细菌群落相对丰度呈极显著正相关关系(P<0.001),是影响细菌群落的最关键土壤因子。此外,碱解氮(P=0.007)、全氮(P=0.004)和阳离子交换量(P=0.003)与土壤细菌群落之间存在显著正相关关系,是影响细菌群落的主要土壤因子。
-
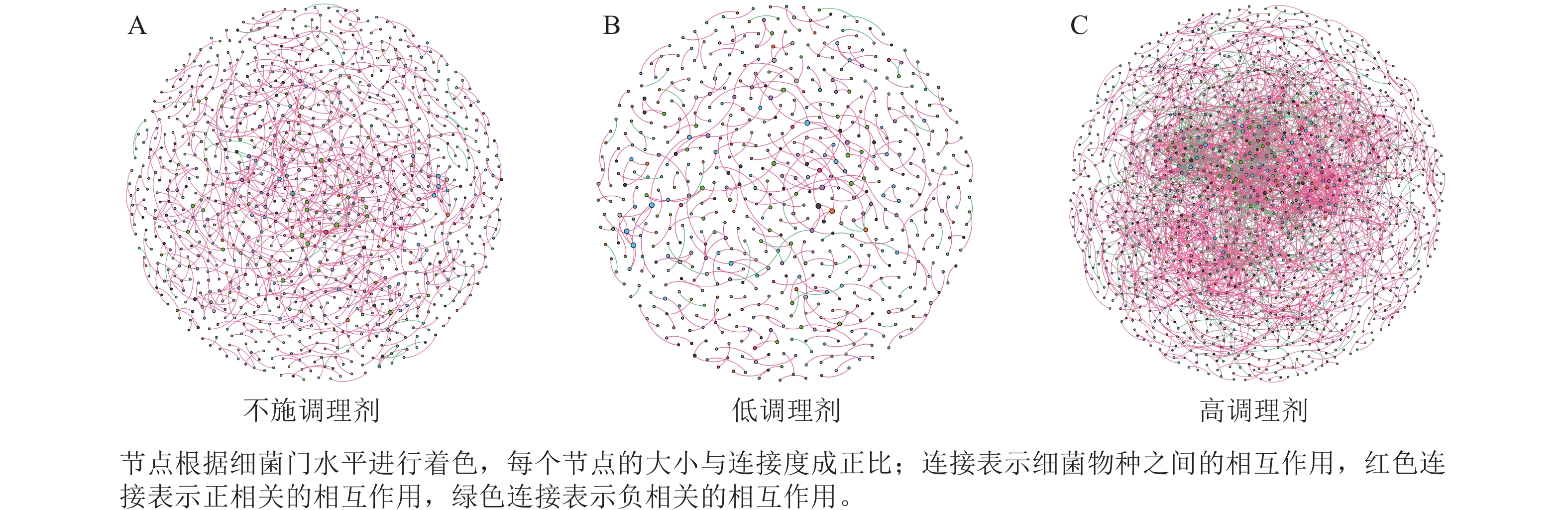
为研究不同土壤调理剂及有机肥施用量对土壤微生物群落的影响,分别构建了2种因素影响的细菌群落共现网络图。不同调理剂施用量处理土壤细菌群落共现网络如图5所示。结果表明:不施调理剂、低调理剂处理下明显降低了网络节点数和边数,但提高了网络模块化程度。高调理剂处理的细菌共现网络具有更高的平均加权度,说明不同调理剂施用量显著影响了土壤细菌共现网络拓扑结构,高调理剂处理的网络结构最复杂,更倾向于一个整体(表3)。相比于不施用有机肥,低有机肥和高有机肥处理均简化了土壤细菌共现网络。有机肥施用量对土壤细菌共现网络也存在显著影响,具体表现为高有机肥处理的土壤细菌共现网络比低有机肥处理更复杂(图6)。由表4可以看出:低有机肥处理的节点数、边数、平均度和平均加权度均为最低值,模块化占比达最高值,说明该处理下,土壤细菌网络虽然整体连接稀疏,种类和相互作用少,但内部结构高度组织化,形成了多个紧密联系的独立模块。
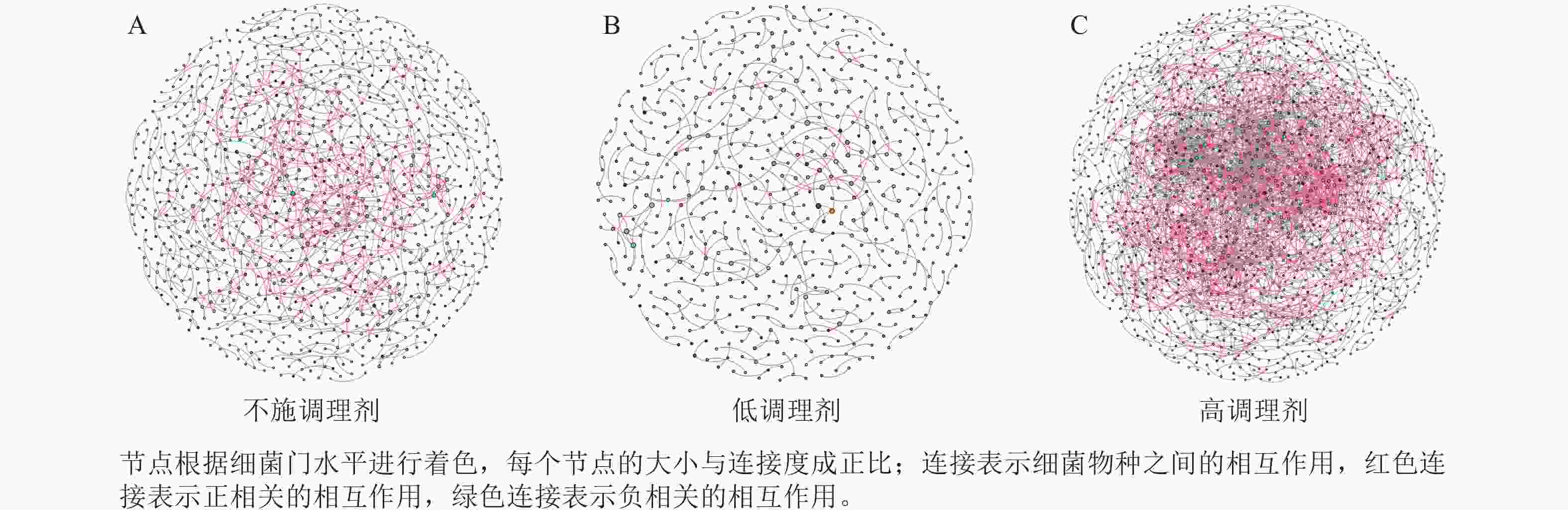
Figure 5. Soil bacteria co-occurrence networks under different application rates of conditioner agents
处理 节点数 边数 平均度 平均
加权度正相关
比率/%负相关
比率/%模块化 不施调
理剂1 012 833 1.646 1.618 95.92 4.08 0.985 低调理
剂处理598 390 1.304 1.285 89.74 10.26 0.993 高调理
剂处理1 424 2 677 3.760 3.657 78.74 21.26 0.826 Table 3. Main topological properties of co-occurrence networks of soil bacterial communities under different application rates of conditioning agents
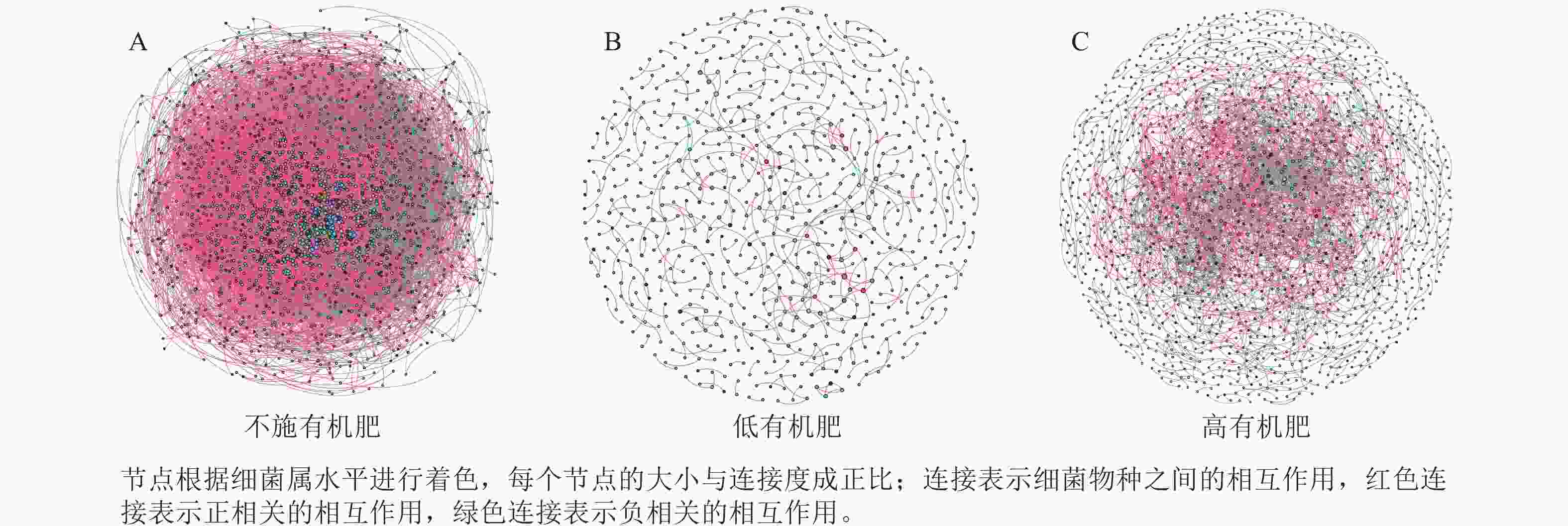
Figure 6. Soil bacterial co-occurrence networks under different application rates of organic fertilizer
处理 节点数 边数 平均度 平均
加权度正相关
比率/%负相关
比率/%模块化 不施
有机肥2 066 52 277 50.607 46.873 91.05 8.95 0.438 低有机
肥处理675 479 1.419 1.397 72.65 27.35 0.983 高有机
肥处理1 502 3 175 4.228 4.109 69.83 30.17 0.784 Table 4. Main topological properties of the co-occurrence network of soil bacterial communities under different application rates of organic fertilizer
-
施肥能够通过改变土壤养分含量影响作物产量和品质,但由于不同肥料的成分差异,对土壤的pH和土壤养分影响不同[18]。作为衡量土壤质量的关键指标,土壤pH对土壤养分有效性、微生物活性及作物生长发育均有着重要影响[19]。黄颖博等[20]研究发现:有机肥中有机质分解产生的碱性物质能中和土壤酸性,提高土壤pH,进而增强土壤生态系统的稳定性。但本研究发现,有机肥施用量对土壤pH无显著影响,仅在不施调理剂处理下,随着有机肥施用量的增加,土壤pH显著上升,这与罗克万等[21]研究结果一致。土壤调理剂对于土壤性质同样具有较强的调节作用。本研究中,不施有机肥处理下随着调理剂施用量的增加,土壤pH显著升高。这是由于调理剂所含的氧化钙、碳酸钙等碱性成分通过酸碱中和反应使土壤pH显著提升。该结果与吴拓铮等[22]的研究结果相似。
土壤有机质和速效养分是衡量土壤肥力的重要指标。本研究发现:随着有机肥施用量的增加,土壤有效磷质量分数显著下降,可能是由于该有机肥为山核桃蒲壳发酵而成,碱性较强,导致土壤磷有效性降低。同时,山核桃蒲壳的吸附性较强,也是导致土壤有效磷降低的可能原因之一[23]。但随着土壤调理剂及有机肥的施用,土壤有机质质量分数也随着显著升高,且调理剂与有机肥的交互作用对土壤有机质和土壤养分有显著影响,说明调理剂施用可能增加了有机质与矿物及土壤的吸附,提高了土壤有机质的稳定性,其与有机肥配施有助于提升土壤有机质质量分数。
-
在农田生态系统中微生物发挥着关键作用,其多样性水平既反映土壤的健康状况也对作物生长产生影响[24]。已有研究表明:不同施肥方式会对耕层土壤细菌群落的丰富度和多样性产生显著影响[25]。于金珠等[26]研究指出,施用调理剂可提高土壤细菌群落的丰富度和多样性。顾美英等[27]研究发现施用有机肥同样起到这种效果。本研究中,不同施肥处理下土壤α多样性并没有显著差异。土壤细菌Shannon指数和Chao 1指数随有机肥施用量的增加有上升趋势,但差异并不显著,而在高调理剂处理下多样性指数却下降,可能是由于高调理剂处理对土壤细菌多样性存在一定的抑制作用。
本研究在门水平的分析显示:放线菌门和变形菌门为土壤细菌中的优势种群,相对丰度分别为20.27%~29.13%和21.80%~26.40%。在施用相同量调理剂处理下,有机肥对细菌种群有显著影响,随着有机肥施用量的递增,放线菌门、酸杆菌门和绿弯菌门的相对丰度随之升高,而厚壁菌门相对丰度则呈下降趋势。这一现象可能是由于放线菌门作为有益菌群,其数量的增加可提升土壤有机质的利用率,进而抑制土传病害,改善土壤生态系统功能[28],而厚壁菌门对环境变化的适应能力较弱,有机肥的施用可能会引入更多有机物质分解者,通过资源竞争抑制厚壁菌群的生长。在施用相同量有机肥处理下,随着调理剂施用量的增加,放线菌门、变形菌门、酸杆菌门和芽单胞菌门相对丰度呈现明显的增长趋势,但在高有机肥处理下相对丰度受到抑制而降低,表明高有机肥处理对微生物群落的影响具有选择性[29]。
在不同调理剂施用量处理下,随着有机肥施用量的增加,甲基杆菌属和分枝杆菌属的相对丰度均显著降低。甲基杆菌属和分枝杆菌属均属于有益菌。有研究表明[30]:甲基杆菌属可通过竞争排斥某些病原菌,减少植物病害发生,进而促进植物对营养物质的吸收。在施用相同量有机肥处理下,甲基杆菌属和分枝杆菌属相对丰度随着调理剂施用量的增加而逐渐降低,而索丝菌属、亚硝化球菌属和鞘氨醇杆菌属相对丰度显著提升。亚硝化球菌属在氮循环过程中具有重要作用,尤其在富含氨的环境中表现活跃。调理剂施用量的增加促进有机肥中铵态氮转化为氨,刺激了亚硝化球菌属的生长。鞘氨醇杆菌属对环境中的污染物具有降解能力,且在碳循环中发挥作用,调理剂施用提高了土壤pH,为其降解有机肥,参与碳循环创造了更适宜的环境条件。
调理剂和有机肥的施用通过影响土壤理化性质,间接对土壤微生物群落组成及生态功能产生影响。本研究发现:放线菌门与土壤pH和速效钾质量分数呈极显著正相关。这类微生物在养分富集环境中具有竞争优势,在有机质分解过程中可能起到关键作用。这些发现不仅证实了“环境选择”理论在土壤微生物群落构建中的重要性,更揭示了特定功能菌群与环境因子之间的协同进化关系。多数细菌门类与pH呈正相关,这与中性环境更有利于微生物多样性这一经典生态学观点一致。这些相关性模式为预测土壤生态系统功能变化提供了重要指标,也为调控土壤微生物群落以改善生态功能提供了理论依据。由图4可知:JABFSM01和盖亚菌属与土壤全氮质量分数呈显著正相关,可能是由于它们在土壤氮循环中扮演着关键角色,包括促进有机氮的矿化,参与固氮作用以及提高氮素转化效率等。这些功能使它们成为土壤肥力的重要贡献者。
-
本研究非度量多维标度分析结果表明:不施调理剂与高调理剂处理之间细菌群落结构存在显著差异,说明施用大量调理剂会使土壤细菌群落结构发生显著改变。冗余分析结果表明:土壤有效磷、碱解氮、全氮质量分数和阳离子交换量是导致土壤细菌群落结构变化的主要环境因子。土壤有效磷和全氮作为关键养分因子,通过氮磷协同效应,增强氮磷循环效率从而改变土壤细菌群落结构。本研究中,高调理剂处理的土壤细菌共现网络结构的节点数、边数、平均度及平均加权度均高于不施调理剂和低调理剂处理,但模块化小于这2个处理,说明施肥措施影响了土壤微生物的共现网络拓扑结构。低调理剂处理下土壤的节点数、边数以及平均加权度低于不施调理剂处理,而模块化高于不施调理剂处理。一方面是由于调理剂本身的酸碱性会影响土壤pH,打破原有菌群对土壤酸碱的平衡作用,低调理剂处理可能会抑制嗜酸菌的生长,从而导致网络复杂度的下降;另一方面,低调理剂的颗粒结构可能短暂改变土壤孔隙分布,导致好氧菌(如变形菌门)的生存空间受限,而好氧菌通常是有机肥分解的关键参与者,其互作关系减少会进一步降低网络边数。这一现象与YUAN等[31]的研究结果一致。低调理剂处理对微环境的影响最终表现为网络节点数与边数的下降。在不同有机肥施用量处理下,施用有机肥明显降低了网络节点和边的数量,但提高了网络模块化程度。不施有机肥的茄土壤细菌共现网络具有更高的网络密度,说明施用有机肥影响了土壤细菌的共现网络拓扑结构。不施有机肥处理网络结构最复杂,更倾向于一个整体。WANG等[32]研究结果表明:不施有机肥土壤微生物网络的节点连通性比施有机肥高29%~34%。此外,CHEN等[33]指出:不施肥处理中腐殖质需依赖3种细菌协同作用,而有机肥易分解碳可被单一菌群利用,切断协同,进而导致不施肥处理细菌共现网络的节点连通性高于有机肥处理。这与本研究结果一致。CHEN等[34]研究发现:低有机肥处理土壤细菌多样性比不施有机肥处理低,且显著低于高有机肥处理,由于低有机肥处理提供的碳源仅支撑优势菌群增殖,从而导致其边数及节点数的降低,稀有细菌因 “碳源获取能力弱+竞争劣势”被大量淘汰;而高有机肥处理的碳源过剩,反而为稀有细菌提供了“拾遗补缺”的生存空间,这直接解释了本研究“低有机肥网络节点数最低”的现象。但高有机肥处理后,有机肥富集多类功能菌,形成协同网络,且有机肥中的有机磷与土壤调理剂带入的钙离子相结合,减少磷的竞争,间接促进网络恢复[9]。由此可以推断,调理剂与有机肥配合施用可通过提高土壤中富营养类群及有益菌的相对丰度,优化土壤细菌群落结构,同时降低土壤中寡营养类群和致病菌的相对丰度,使土壤细菌群落结构变得更稳定、紧密。综上所述,从细菌共现网络的角度来说,相比于低调理剂和有机肥处理,高调理剂和有机肥处理能更好地提高细菌网络稳定性,优化网络结构。
-
调理剂和有机肥施用量显著影响土壤pH、有机质和速效养分等化学性质。与不施调理剂处理相比,高调理剂处理显著提高了土壤养分质量分数;高有机肥处理的土壤速效钾和有效磷质量分数随调理剂施用量增加显著提高。
在相同有机肥施用处理下,不同调理剂施用量显著改变了细菌群落结构。土壤有效磷、碱解氮、全氮和阳离子交换量是影响土壤细菌群落结构的主要环境因子。高调理剂处理的土壤细菌共现网络比不施调理剂和低调理剂处理的网络更完整;与低有机肥处理相比,高有机肥处理提高了细菌共现网络的复杂度。
总体来看,土壤调理剂和有机肥施用均能改善土壤理化性质,但从细菌群落的角度来看, 1.5 t·hm−2调理剂和4.5 t·hm−2有机肥处理能明显优化细菌群落共现网络,提升土壤健康水平。
Short-term response characteristics of soil bacterial community in open-field vegetable soils to combined application of organic fertilizer and soil conditioner
doi: 10.11833/j.issn.2095-0756.20250403
- Received Date: 2025-07-30
- Accepted Date: 2026-03-30
- Rev Recd Date: 2026-03-26
-
Key words:
- open-field vegetables /
- soil conditioner /
- organic fertilizer /
- soil bacterial community
Abstract:
| Citation: | SHI Luyi, XU Rui, WU Qifeng, et al. Short-term response characteristics of soil bacterial community in open-field vegetable soils to combined application of organic fertilizer and soil conditioner[J]. Journal of Zhejiang A&F University, 2026, 43(X): 1−12 doi: 10.11833/j.issn.2095-0756.20250403 |









 DownLoad:
DownLoad: