-
植物内生真菌(endophytic fungi)是指那些全部或近整个生命周期生活在宿主植物的各类细胞及细胞间隙、组织、器官内部,却不会使寄主表现出不良症状的真菌类群[1-2]。大量研究表明:内生真菌可以通过竞争生存空间和营养元素[3]、溶菌作用[4]、影响病原真菌菌丝生长及孢子发育方式[5]等抑制病原菌的生长,同时刺激根毛形成和根茎生长,提高植物感病时的光合作用及酶活性[6-7],提高植物抗病能力。沙地云杉Picea mongolica是松科Pinaceae云杉属Picea常绿针叶乔木,是内蒙古特有树种,沙质土地上的活化石[8]。由于近年来大面积的立枯病等病虫害的频繁发生[9],沙地云杉正面临着可能灭绝的危险。目前,喷施化学农药是防治严重林木病害的主要措施,但农药利用率低下,极不适宜应用于大面积的森林,急需开发天然生物资源代替传统化学农药试剂。本研究选取前期筛选的4株具有强拮抗作用的沙地云杉内生真菌为试验材料,以沙地云杉立枯病病原真菌Fusarium sp.为靶标菌,采用体外对峙和盆栽试验,探究内生拮抗菌的拮抗活性和对沙地云杉的抗病作用,旨在为利用生物资源、开发生防菌提供依据。
HTML
-
4株沙地云杉内生拮抗菌由内蒙古农业大学林学院森林病理实验室提供[10],分别是互隔链格孢菌Alternaria alternata (SDYS36)、三线镰刀菌Fusarium tricinctum (SDYS63)、产黄青霉菌Penicillium chrysogenum (SDYS95)、聚多曲霉菌Aspergillus sydowii (SDYS180)。
-
沙地云杉立枯病病原菌Fusarium sp.由内蒙古农业大学林学院森林病理实验室提供。
-
沙地云杉种子由赤峰市白音敖包自然保护区提供。除杂后挑选种实饱满、色泽正常的健康种子,低温保存备用。千粒重为0.011 4 g。
-
马铃薯葡萄糖琼脂(PDA)固体培养基、马铃薯葡萄糖(PD)液体培养基。
-
在超净工作台内将沙地云杉内生拮抗菌和沙地云杉立枯病病原菌供试菌株切成0.2 cm×0.2 cm的菌块,接种于已灭菌的PDA平板进行纯培养;无菌条件下,用5 mm打孔器在培养7 d的供试菌株平板边缘打成圆形菌饼,接种于加有100 mL灭菌后的PD液体培养基的250 mL三角瓶中,震荡培养5~7 d (28 ℃,180 r·min−1),得到发酵菌液备用;取上述发酵液离心(8 000 r·min−1,4 ℃) 2 min后留上清液,在超净工作台通过0.22 µm滤膜,即为无菌发酵液。
-
采用生长速率法[11],以立枯病病原菌为靶标菌,测试沙地云杉内生拮抗菌株发酵液拮抗活性。无菌条件下,加定量无菌发酵液于PDA培养基(40~50 ℃)混匀,制成平板(Ф=7.5 cm),冷凝后在中心位置接入靶标菌,每组均设5个重复,以加入相同比例无菌水的平板上靶标菌的生长作为对照(ck),25 ℃倒置培养。待对照中靶标菌长满平板时,测量靶标菌的菌落直径,计算相对抑菌率。相对抑菌率RI=(Rc−Rp)/Rc×100%。Rc为对照靶标病原菌的菌落直径,Rp为对峙实验中靶标病原菌的菌落直径。
-
采用皿内对峙法[12],以立枯病病原菌为靶标菌,测试沙地云杉内生拮抗菌株拮抗活性。无菌条件下,将内生拮抗菌株和靶标菌分别接种在PDA平板(Ф=7.5 cm)上,各距中心位置2.5 cm,以单独培养靶标菌为对照(ck),均设5个重复,25 ℃倒置培养。记录对峙培养中靶标菌菌落的相对生长距离,计算相对抑菌率。
-
采用对扣抑制法[13],以立枯病病原菌为靶标菌,测试沙地云杉内生拮抗菌株挥发性代谢产物的拮抗活性。无菌条件下,将内生拮抗菌株和靶标菌分别接种在2个PDA平板(Ф=9.0 cm)中心,撤去皿盖,靶标菌在上、拮抗菌在下对扣放置,以单独培养靶标菌为对照(ck),均设5个重复,25 ℃恒温培养。记录对峙培养中靶标菌菌落的相对生长距离,计算相对抑菌率。
-
参照袁宗胜等[14]、邓勋等[15]的方法及《植物生理学实验指导》[16],通过灌根处理将内生拮抗菌株及发酵菌液接种在外观大小相似的1年生沙地云杉健壮幼苗上,并设置单独浇灌PD液体培养基(PD)和清水(ck)等2组对照,每种处理设置苗200株,共有苗1 200株,待土壤表面干燥后接种沙地云杉立枯病原菌。发病后,比较病苗生长30和45 d时的形态(株高生长量、地径生长量、根长生长量)和叶片组织的可溶性蛋白质质量分数、丙二醛质量摩尔浓度、保护酶活性的变化,分析内生拮抗菌株对沙地云杉染病幼苗抵抗立枯病的影响。
1.1. 材料
1.1.1. 供试沙地云杉内生拮抗菌
1.1.2. 供试林木病原真菌
1.1.3. 供试沙地云杉种子
1.1.4. 主要培养基
1.2. 方法
1.2.1. 菌株培养
1.2.2. 内生拮抗菌发酵液对沙地云杉立枯病病原菌的拮抗活性测定
1.2.3. 内生拮抗菌菌体对沙地云杉立枯病病原菌的拮抗活性测定
1.2.4. 内生拮抗菌挥发性代谢产物对沙地云杉立枯病病原菌的拮抗活性测定
1.2.5. 沙地云杉内生拮抗菌对盆栽沙地云杉立枯病拮抗试验
-
4株内生真菌的发酵液对病原菌均有显著的抑制作用,抑菌率为51.64%~78.67%。4株内生真菌对沙地云杉立枯病病原菌的抑菌率均在51.64%以上。SDYS180发酵液的抑菌率最高,达到78.67%。与另外3株内生真菌发酵液间差异显著(P<0.05) (表1)。4株真菌发酵液的抑菌率从大到小依次为SDYS180、SDYS36、SDYS63、SDYS95。
菌株编号 抑菌率/% 发酵液 菌体 挥发性代谢产物 SDYS36 70.67±3.52 b 55.24±0.47 c 16.11±2.64 ab SDYS63 60.44±2.78 c 51.96±0.08 d 16.94±2.29 a SDYS95 51.64±0.43 d 69.64±0.31 a 13.06±2.63 b SDYS180 78.67±1.34 a 66.27±0.69 b 15.00±1.11 ab 说明:数据为平均值±标准差,不同小写字母表示不同内生拮抗
菌种在0.05水平上差异显著Table 1. Antagonistic effects of endophytic fungi in P. mongolica on Fusarium sp.
-
4株沙地云杉内生真菌菌体本身对沙地云杉立枯病病原真菌均有显著的抑制作用,抑菌率均大于51.96%。抑菌率从大到小依次为SDYS95、SDYS180、SDYS36、SDYS63。其中,SDYS95的抑制效果最强,抑菌率达到69.64%,与另外3株内生真菌差异显著(P<0.05) (表1)。
-
4株沙地云杉内生真菌菌株的挥发性代谢产物对沙地云杉立枯病病原真菌的生长有一定的拮抗作用,但抑制率较低,抑菌率从大到小依次为SDYS63、SDYS36、SDYS180、SDYS95 (表1)。其中,拮抗效果最强的SDYS63,抑菌率仅达到16.94%,与SDYS95内生真菌差异显著(P<0.05);拮抗效果最弱的是SDYS95,抑菌率仅有13.06%。
-
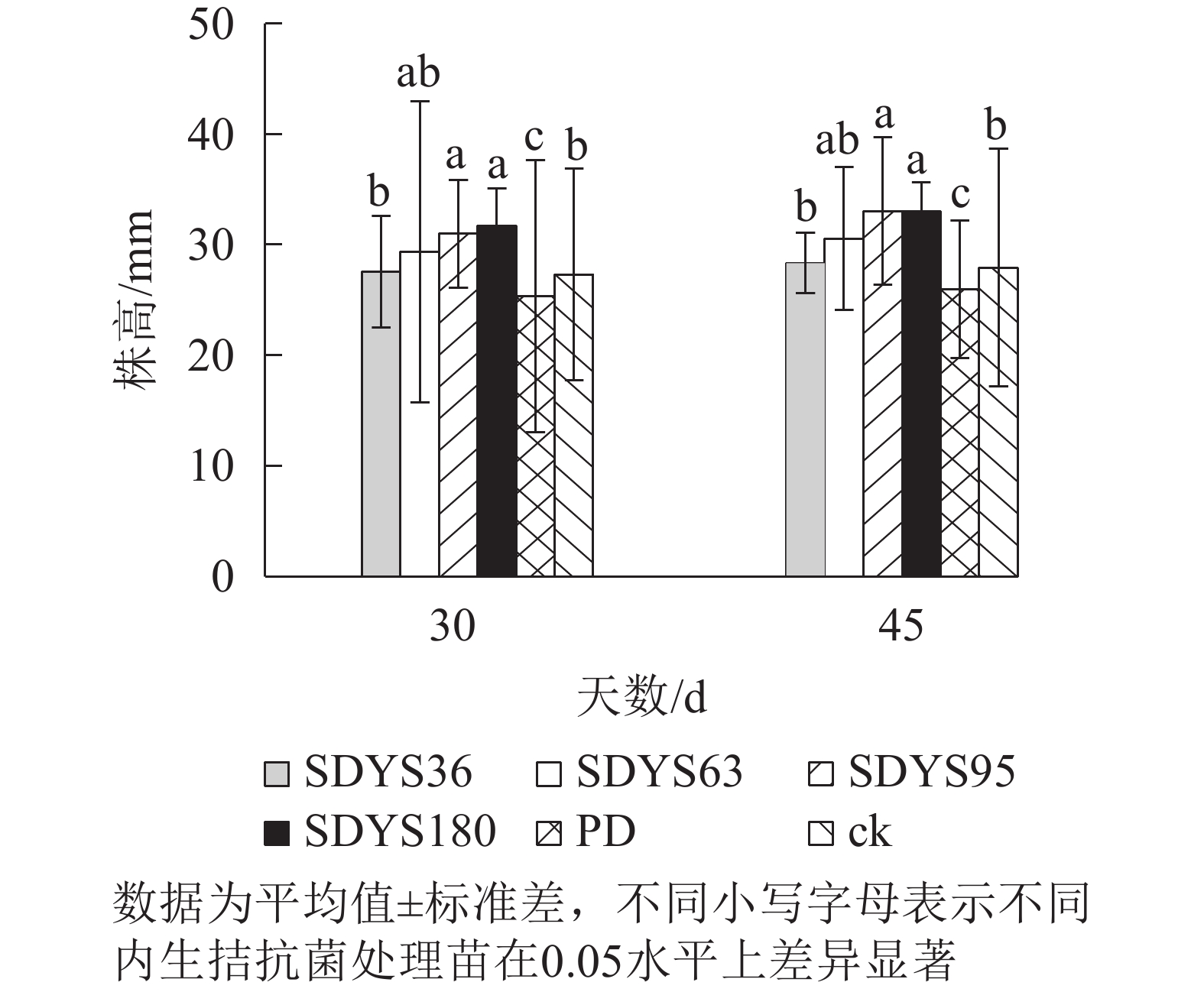
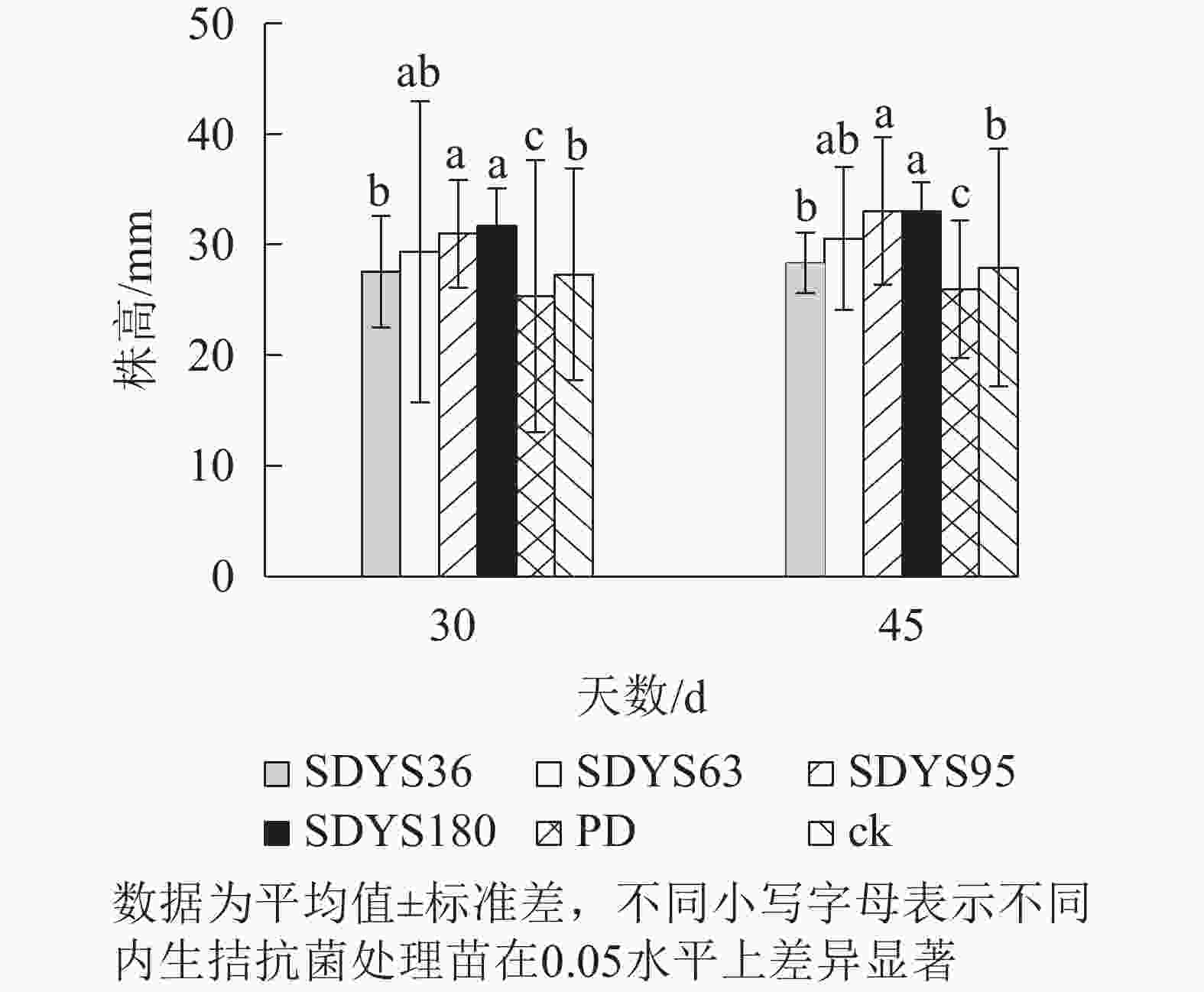
发病30 d时,各处理染病幼苗株高从高到低依次表现为SDYS180、SDYS95、SDYS63、SDYS36、ck、PD,试验组株高较ck高0.91%~16.01%,PD生长势衰弱,可见菌丝着生。发病45 d时,各处理染病幼苗株高从高到低依次为SDYS180、SDYS95、SDYS63、SDYS36、ck、PD,试验组苗木的株高较ck高出1.42%~18.31%,ck叶片退绿枯黄或死亡。通过PD与ck的对比,说明病原菌抑制病苗株高生长;试验组相对ck株高显著增加,说明内生真菌能有效抑制病原菌。2次取样SDYS180的病苗株高生长均最高,与ck和PD具有显著差异(P<0.05)。菌株SDYS180及其代谢产物在苗木株高生长上抗病效果最佳(图1)。
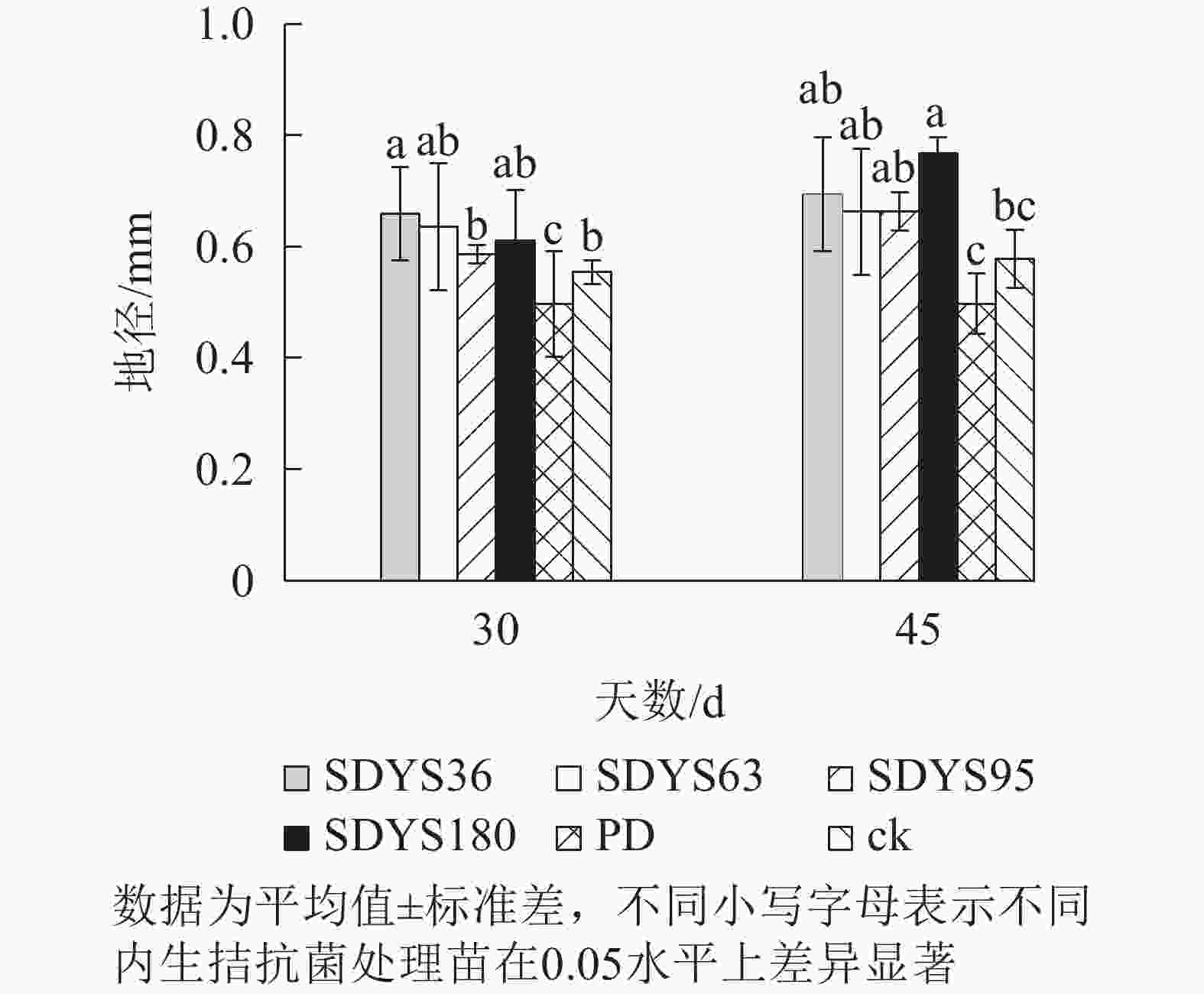
30 d时,各处理染病幼苗地径从大到小依次为SDYS36、SDYS63、SDYS180、SDYS95、ck、PD,试验组地径较ck高5.90%~19.04%,PD较ck低10.22%。45 d时,各处理地径从大到小依次呈现为SDYS180、SDYS36、SDYS95、SDYS63、ck、PD,带菌植株地径较ck高出14.59%~32.64%,PD较ck低13.96%。通过PD与ck的对比,说明病原菌抑制植株地径的增长。试验组相对ck显著增加,说明内生真菌有效抑制发病;2次取样SDYS180的增加幅度最大,与ck、PD差异性均达显著水平(P<0.05),说明菌株SDYS180及其代谢产物在苗木地径生长上抗病效果最佳(图2)。
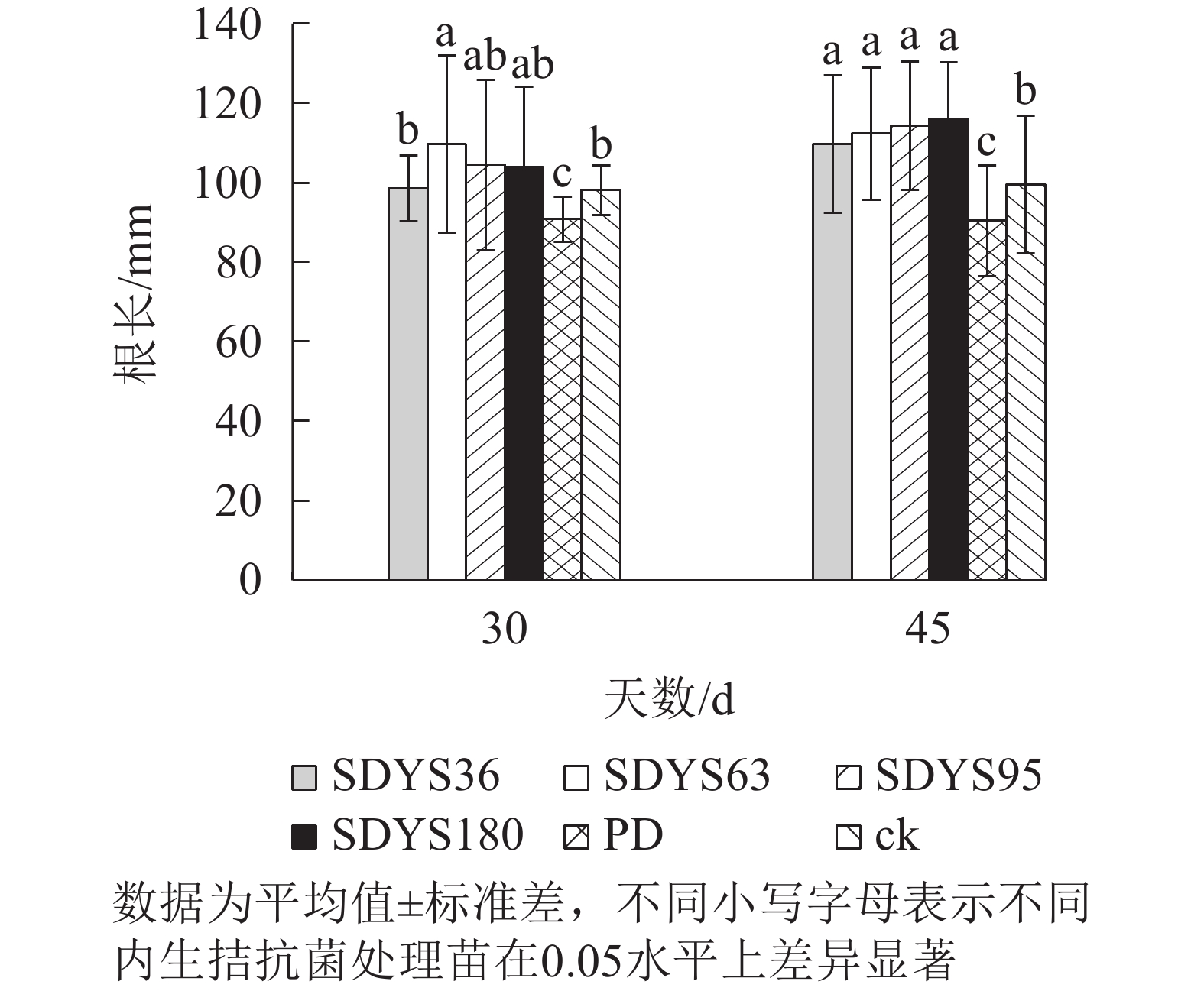
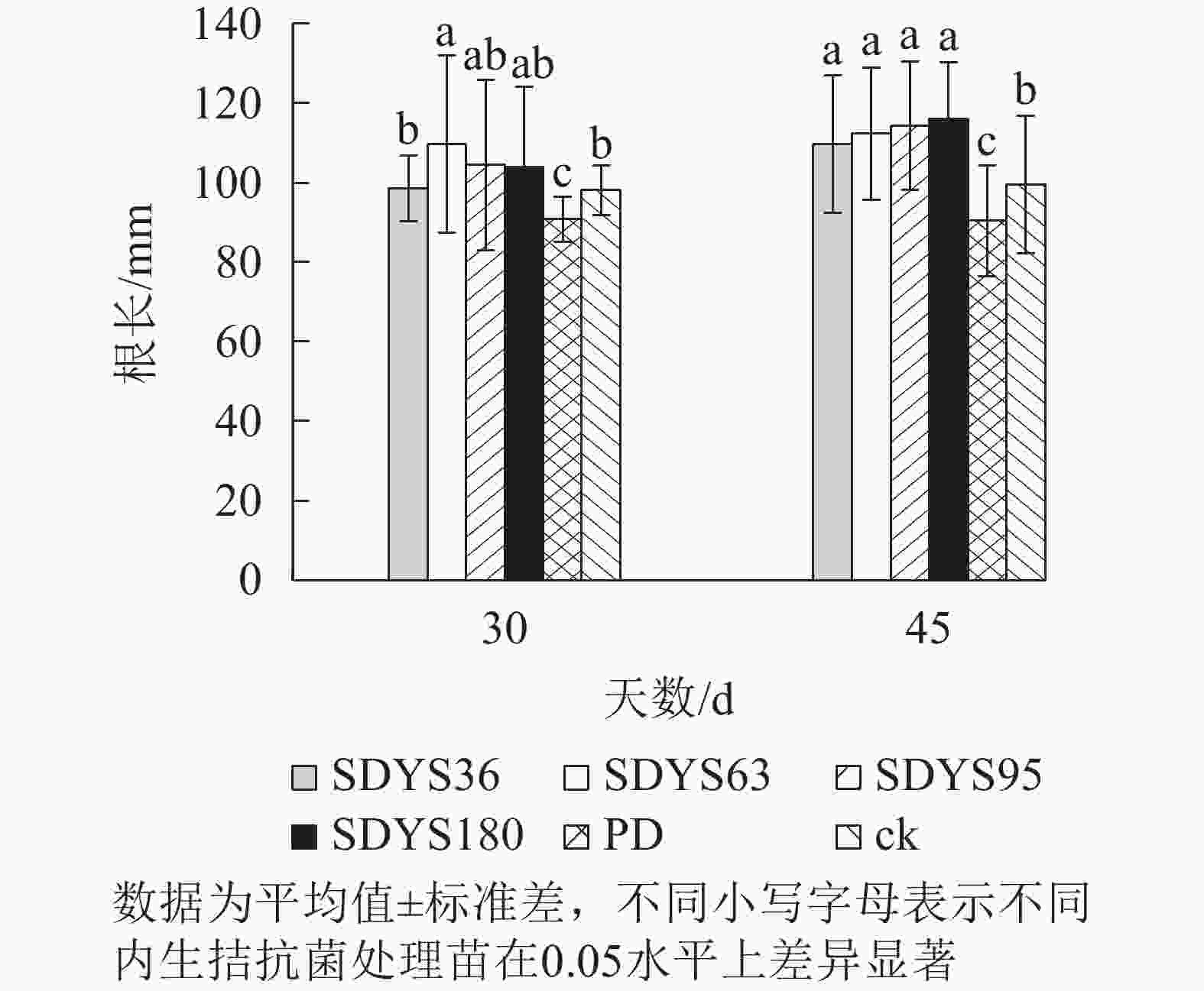
30 d时,各处理染病幼苗根长从大到小依次为SDYS63、SDYS95、SDYS180、SDYS36、ck、PD(图3),试验组根长较ck高0.42%~11.91%,PD较ck低10.22%。45 d时,各处理染病幼苗根长从大到小依次为SDYS180、SDYS95、SDYS63、SDYS36、ck、PD,带菌植株较ck高10.17%~16.56%,PD较ck低13.96%。通过PD与ck的对比,说明病原菌抑制植株根长生长。试验组相对ck显著增加,说明内生真菌有效抑制发病;2次取样SDYS180的根长增加幅度最大,45 d取样与ck、PD差异性均显著(P<0.05),说明菌株SDYS180及其代谢产物在苗木根长生长上抗病效果最佳。
-
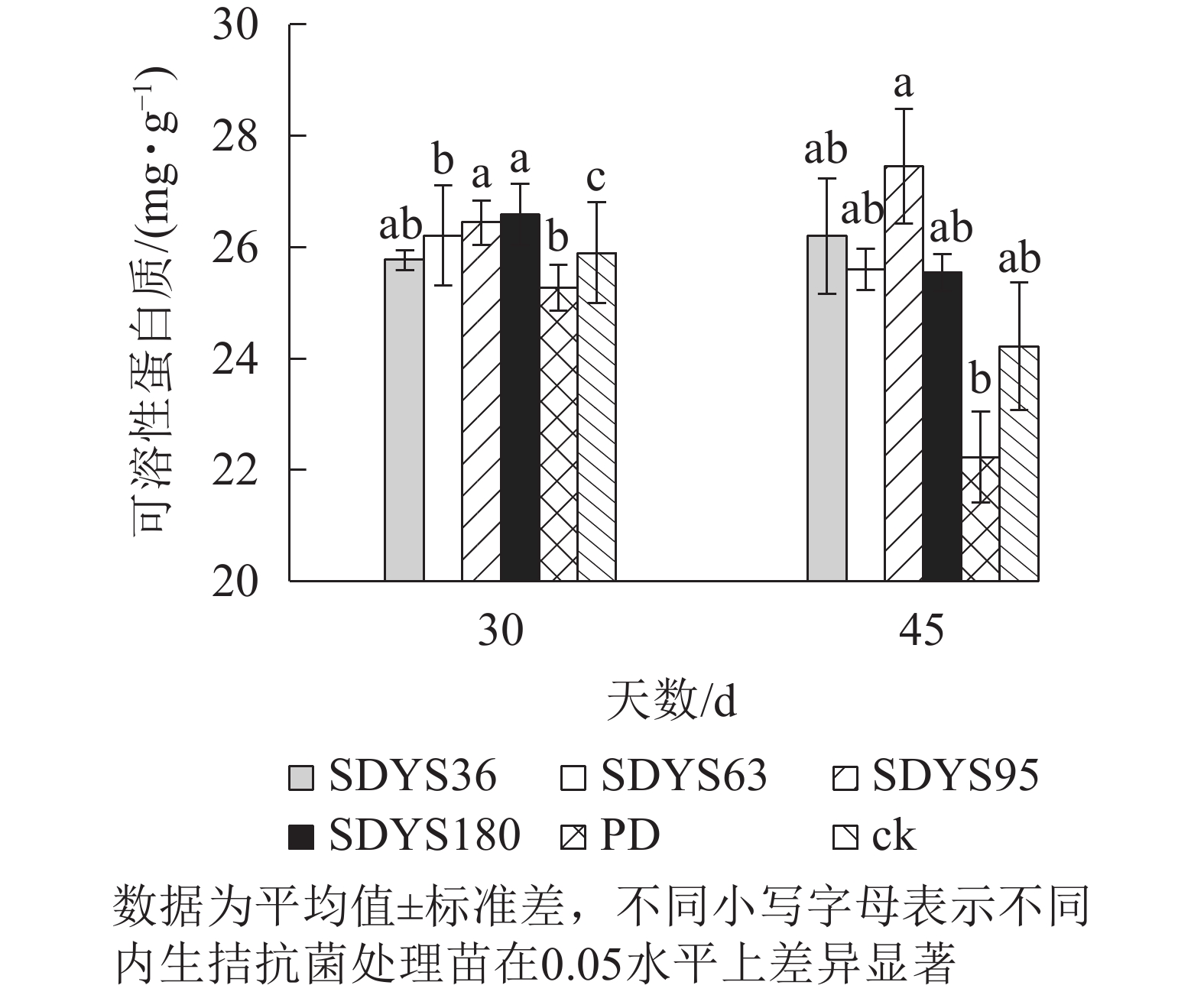
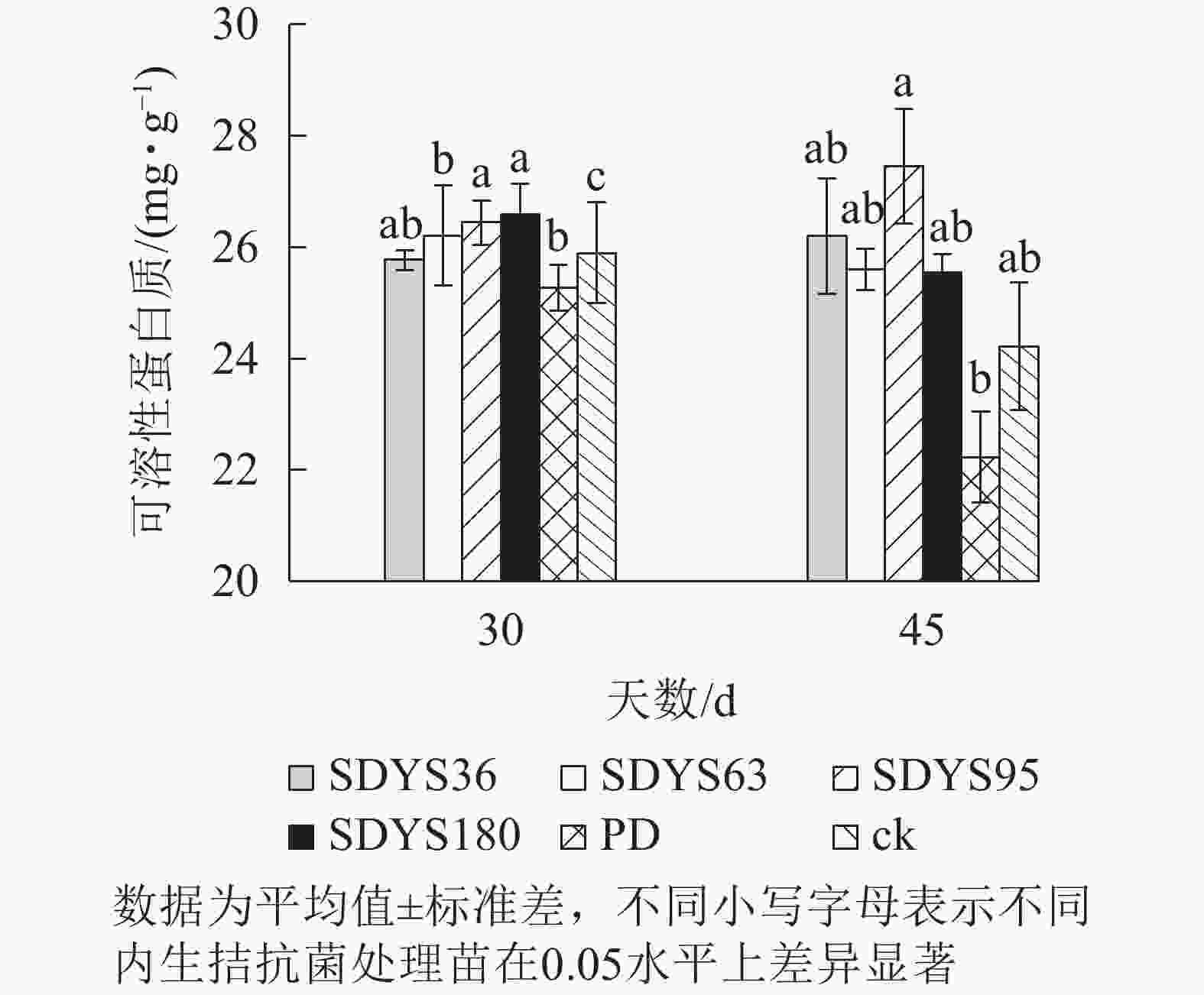
30 d时,各处理染病幼苗中可溶性蛋白质质量分数从高到低依次为SDYS180、SDYS95、SDYS63、ck、SDYS36、PD,SDYS180和SDYS95与ck、PD间差异性显著(P<0.05),高出ck 2.68%和2.10%,PD较ck低2.43%(图4)。45 d时各处理染病幼苗中可溶性蛋白质质量分数从高到低依次为SDYS95、SDYS36、SDYS63、SDYS180、ck、PD,试验组较ck高5.48%~13.41%,PD较ck低8.20%。2次取样PD和ck叶片组织中可溶性蛋白质质量分数大幅下降,病原菌破坏植物细胞结构,影响渗透调节作用。SDYS180、SDYS95、SDYS63相对ck可溶性蛋白质质量分数显著增加,说明3株对应内生真菌有效抑制病原菌对渗透调节的破坏。其中SDYS95的增加幅度最大,说明菌株SDYS95及其代谢产物在苗木上抗病效果最佳,促进植物组织中可溶性蛋白质质量分数的提高。
-
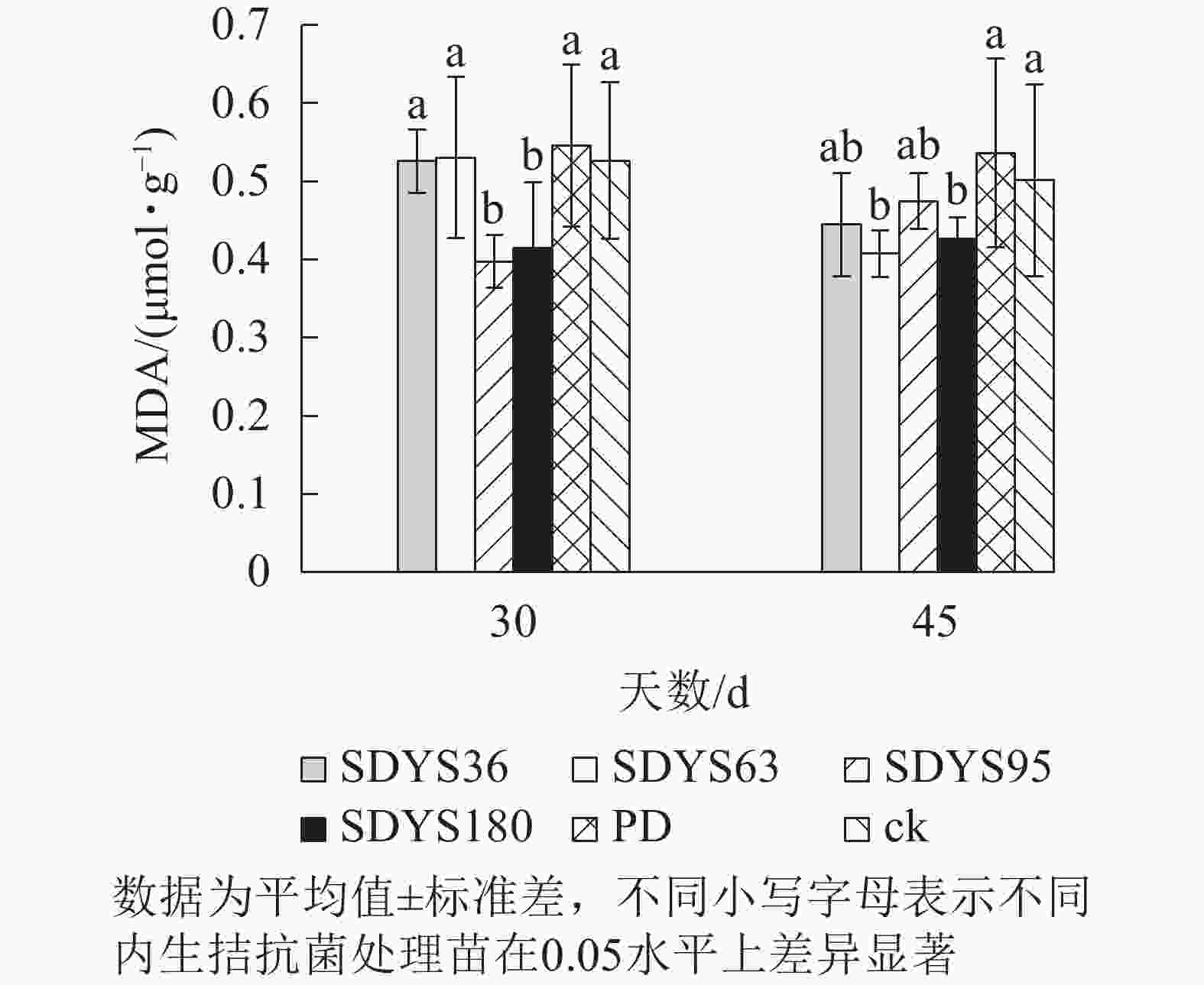
丙二醛(MDA)是由于植物膜脂过氧化作用的重要产物。丙二醛质量摩尔浓度越高,说明植物细胞膜受损越严重。30 d时,各处理染病幼苗中丙二醛质量摩尔浓度从大到小依次为PD、SDYS63、ck、SDYS36、SDYS180、SDYS95,SDYS95和SDYS180较ck降低了24.46%和21.10%;SDYS36降低不明显,和ck仅差0.05%;SDYS63较ck高0.08%,效果不明显;PD较ck高3.70%。45 d时,丙二醛质量摩尔浓度从大到小依次为PD、ck、SDYS95、SDYS36、SDYS180、SDYS63;试验组和对照组比较,丙二醛质量摩尔浓度下降,说明内生真菌抑制病原菌的伤害。其中菌株SDYS63在30 d样品中高于ck,但在45 d样本中丙二醛质量摩尔浓度最低,说明SDYS63增强苗木抗病性,有效减少丙二醛质量摩尔浓度,但拮抗效果不稳定;在2次样品中SDYS180和SDYS95的丙二醛质量摩尔浓度均低于ck,但SDYS180在45 d较SDYS95低10.19%,说明菌株SDYS180拮抗效果良好,而SDYS95作用效果最佳(图5)。
-
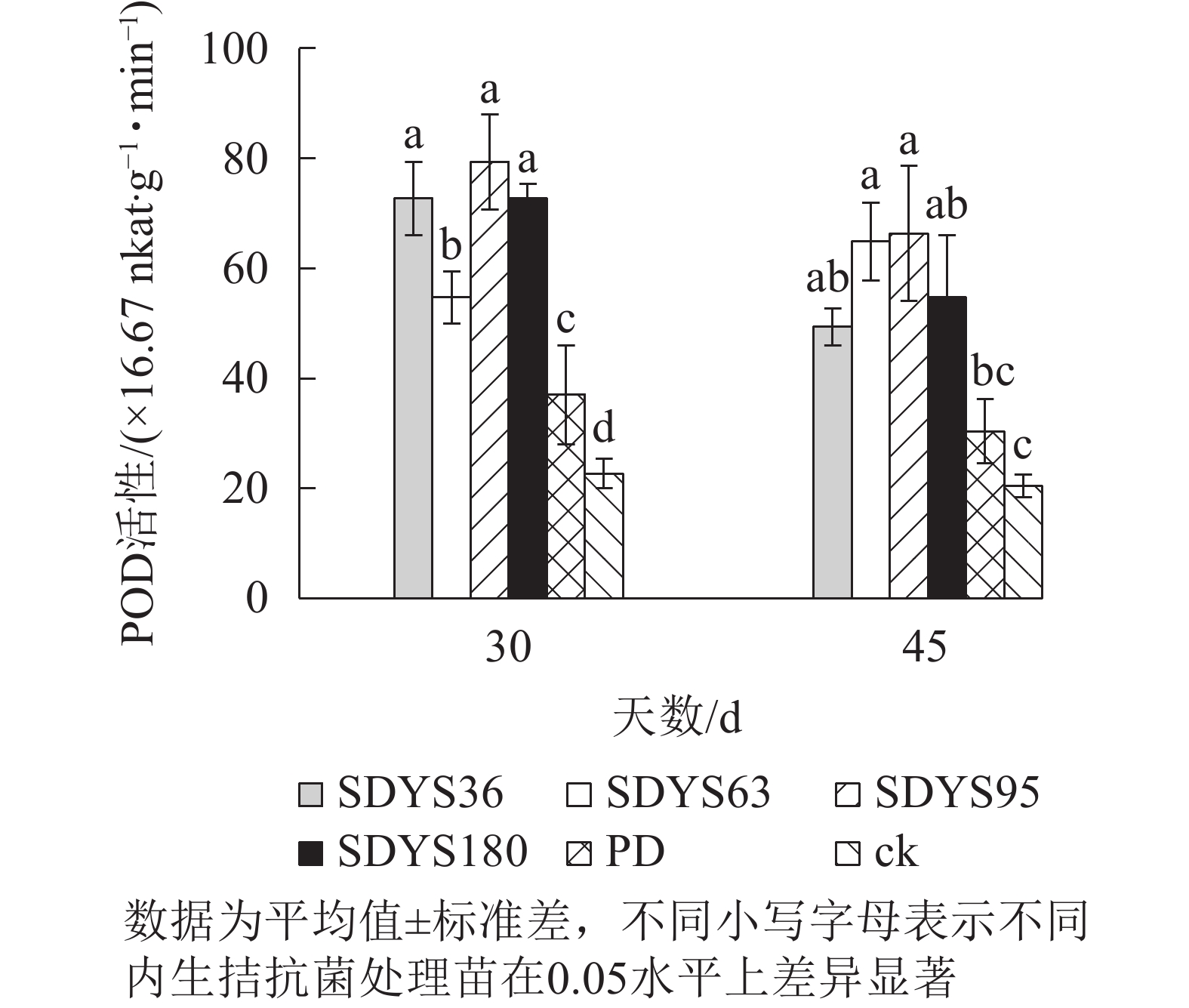
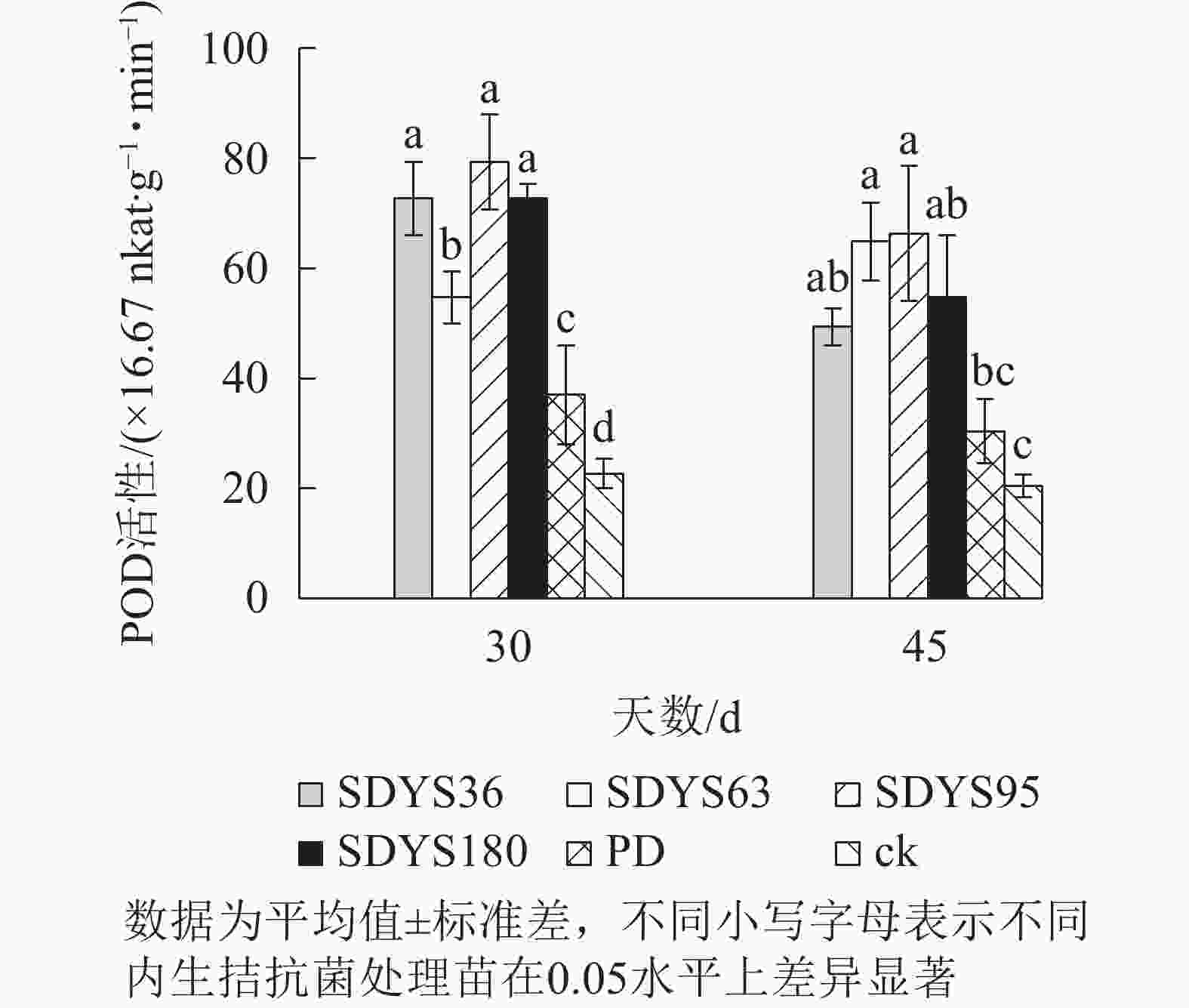
30 d时,各处理染病幼苗POD活性从高到低依次为SDYS95、SDYS180和SDYS36、SDYS63、PD、ck,其中PD较ck高63.24%,试验组较ck高141.18%~250.00%;45 d时,各处理染病幼苗POD活性从高到低依次为SDYS95、SDYS63、SDYS180、SDYS36、PD、ck,其中PD较ck高48.37%,试验组较ck高141.13%~224.46%(图6)。通过PD和ck的比较,说明病原菌对幼苗POD影响不大,反而可能刺激POD活性。在拮抗过程中,在一定程度上被内生真菌抑制。在2次样本中,SDYS95的POD活性均最高,说明菌株SDYS95对提高幼苗抗病性最佳。
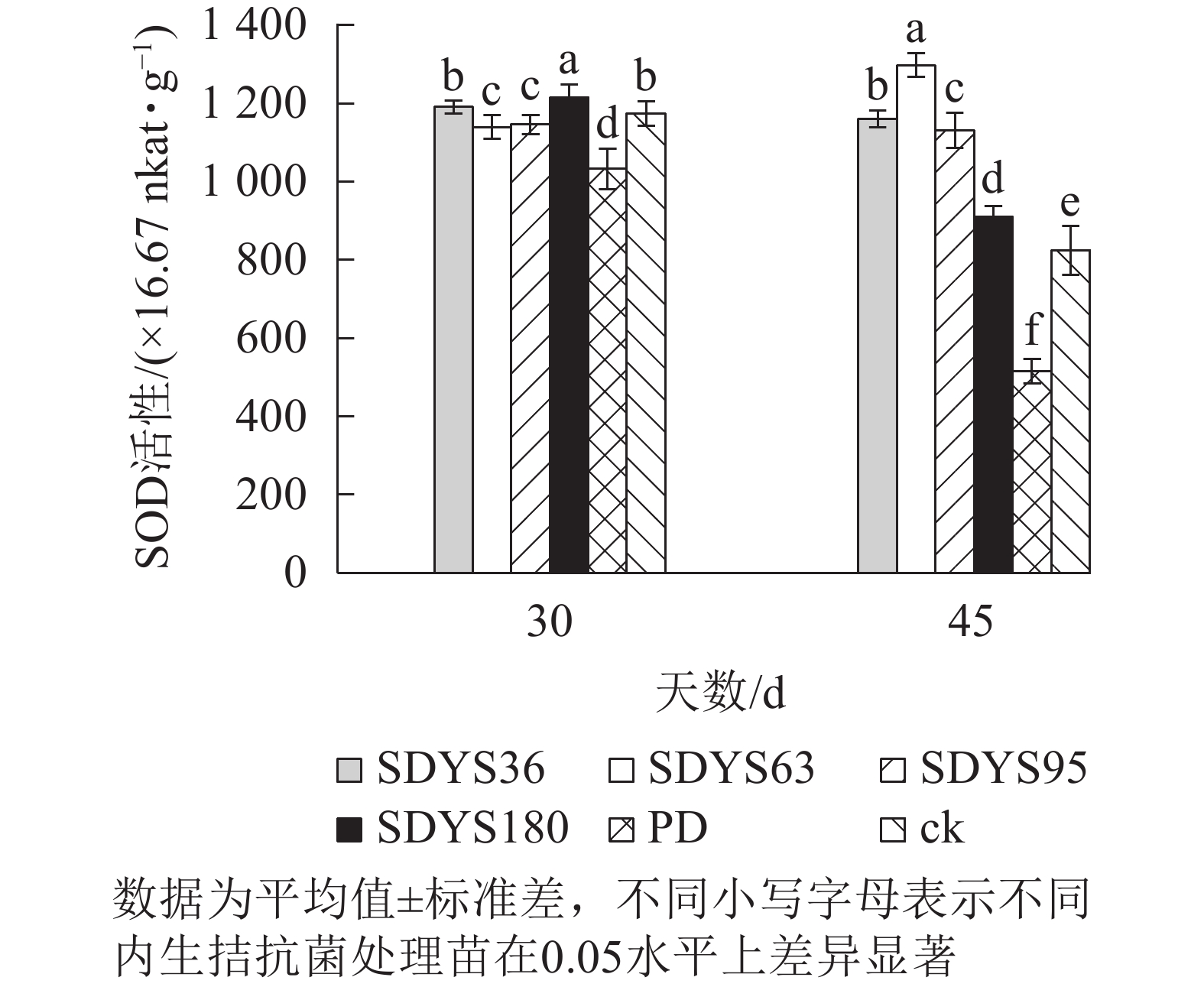
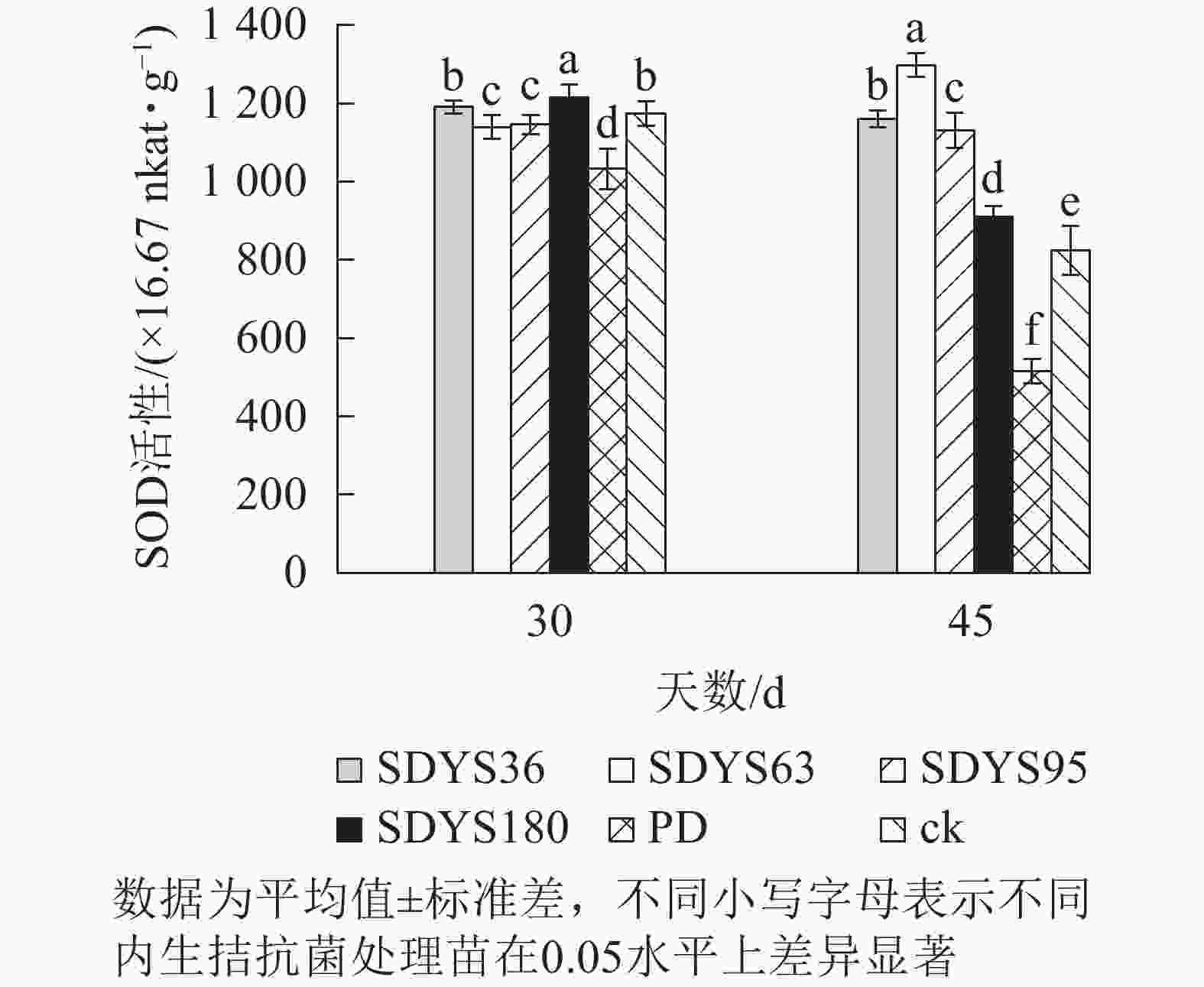
30 d时,各处理染病幼苗SOD活性从高到低依次为SDYS180、SDYS36、ck、SDYS95、SDYS95、PD,其中PD较ck低12.01%,SDYS180较其他处理表现出显著差异(P<0.05),较ck高3.57%;45 d时,各处理染病幼苗SOD活性从高到低依次为SDYS63、SDYS36、SDYS95、SDYS180、ck、PD,其中PD较ck低37.56%,试验组较ck高10.32%~57.36%(图7)。通过PD和ck的比较,说明病原菌对幼苗SOD活性具有抑制作用。在2次样本中,SDYS95的SOD活性均高于ck,说明菌株SDYS95及其代谢产物稳定提高幼苗抗病性;SDYS63在30 d时抑制效果稍弱,但在45 d时抑制效果大幅增加,表明菌种SDYS63的代谢产物的拮抗效果最佳,同时促进SOD的活性增加。
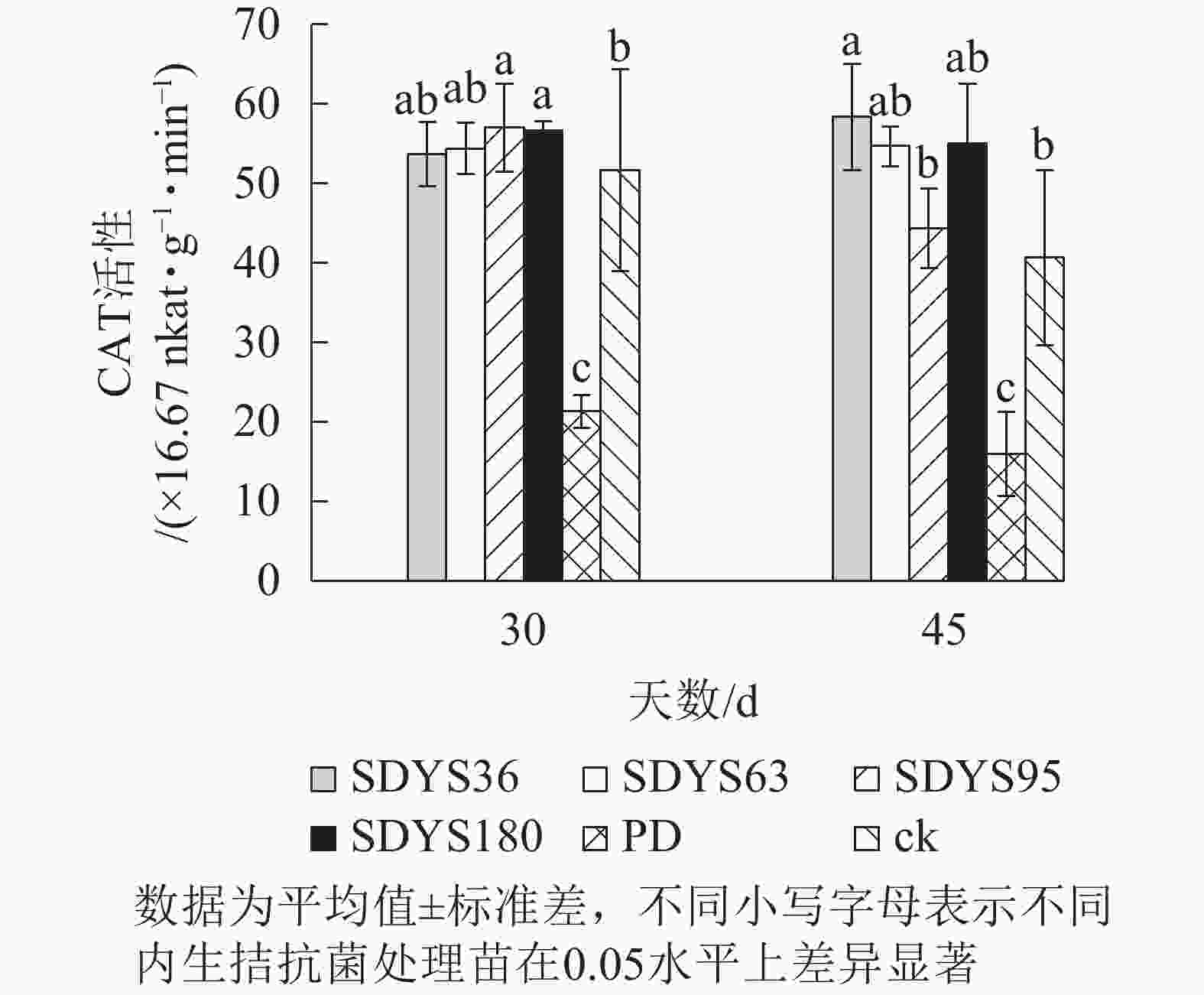
30 d时,各处理染病幼苗CAT活性从高到低依次为SDYS95、SDYS180、SDYS63、SDYS36、ck、PD,其中PD较ck低58.71%,试验组较ck高3.87%~10.322%;45 d时,各处理染病幼苗CAT活性从高到低依次为SDYS36、SDYS180、SDYS63、SDYS95、ck、PD,其中PD较ck低60.66%,试验组较ck高9.02%~43.44%(图8)。通过PD和ck的比较,说明病原菌致病严重,对幼苗CAT活性具有抑制作用,在拮抗过程中,被内生真菌抑制。在30 d样本中,SDYS95的CAT活性最高,说明菌株SDYS95抗病效果良好,可提高幼苗抗病性;SDYS36在30 d时抑制效果稍低,但在45 d时抑制效果最佳,表明菌种SDYS36的代谢产物的拮抗效果良好,同时促进CAT活性增加。
2.1. 4株沙地云杉内生拮抗菌株的体外拮抗活性
2.1.1. 内生拮抗菌株发酵液对沙地云杉立枯病病原菌的拮抗活性
2.1.2. 内生拮抗菌株对沙地云杉立枯病病原菌的拮抗活性
2.1.3. 内生拮抗菌株挥发性代谢产物对沙地云杉立枯病病原菌的拮抗活性
2.2. 4株沙地云杉内生拮抗菌株对盆栽沙地云杉立枯病的拮抗作用
2.2.1. 内生拮抗菌对染病幼苗形态指标的影响
2.2.2. 内生拮抗菌对染病幼苗渗透调节(可溶性蛋白质)的影响
2.2.3. 内生拮抗菌对染病幼苗丙二醛质量摩尔浓度的影响
2.2.4. 内生拮抗菌对染病幼苗保护酶[过氧化物酶(POD)、超氧化物歧化酶(SOD)、过氧化氢酶(CAT)]活性的影响
-
内生真菌作为一类新型待开发的微生物资源,具有广阔的应用前景。目前,开展最广泛的研究领域就是内生真菌的拮抗作用[17]。大量研究表明:内生真菌对多种病原真菌具有很强的拮抗作用和广谱性,通常表现在对生存空间和营养资源的竞争,并产生拮抗活性物质抑制病原菌的生长[18]。本研究结果表明:4种沙地云杉内生真菌经体外培养后,其菌体本身及其代谢产物对植物病原真菌均表现出较强的拮抗活性。但这种拮抗作用存在一定的选择性,可能由不同原因造成。如青霉Penicillium和曲霉属Aspergillus真菌具有更快的扩展速度,在占据生存空间上具有很大的优势;虽然镰刀菌Fusarium本身生长受到限制,但是可以通过产生具有拮抗活性的代谢产物,抑制病原真菌的生长。樊苗苗等[19]对樟叶越桔Vaccinium dunalianum叶中产黄青霉P. chrysogenum、曲霉、链格孢菌Alternaria、镰刀菌等内生真菌及其代谢产物的拮抗活性进行研究,其结果与本研究一致。这些菌株及其代谢产物对病原真菌具有很好的拮抗活性。
-
林木病原真菌可以破坏植物细胞壁,产生有毒物质抑制寄主生长。植物内生真菌及其代谢产物可以有效抑制植物病原菌在寄主植物中的萌发和生长,提高植物感病后的保护酶活性,从而有效提高植物的抗病能力[20]。本研究中,接种沙地云杉内生真菌的感病植株生长幅度均高于对照,说明内生真菌可以有效抑制沙地云杉染病植株上的病原菌,提高植株抗病能力,促进植物组织快速生长。此结果与邓勋等[15]的木霉菌Trichoderma防治松苗立枯病的研究结果一致。
接种沙地云杉内生真菌的感病植株丙二醛质量摩尔浓度显著低于对照组。丙二醛是一种由于质膜过氧化作用而产生的错误的代谢产物,其含量越低,表示细胞膜通透性越低,即受损程度越弱。本研究结果表明:沙地云杉内生真菌可以有效降低组织中丙二醛质量摩尔浓度,有效保护细胞膜。本研究结果与张晶晶等[21]的研究结果一致。
接种沙地云杉内生真菌的感病植株中的过氧化氢酶、过氧化物酶、超氧化物歧化酶的活性比对照表现出不同程度提升。3种酶是植物细胞膜保护酶,起到清除活性氧的重要作用。本研究结果表明:内生真菌可以有效激发沙地云杉抗氧化酶(保护酶)的活性。余承忠等[22]研究认为:病原菌致病性与内生真菌提高寄主抗病能力显著相关。这与本研究结果一致。
可溶性蛋白质既是重要的营养物质,更是关键的渗透调节物质。本次研究结果表明:沙地云杉内生真菌可以增加可溶性蛋白质质量分数,调节组织细胞渗透调节作用。此结果与MA等[23]的研究结果一致。
3.1. 4株沙地云杉内生拮抗菌株体外拮抗活性
3.2. 4株沙地云杉内生拮抗菌株对盆栽沙地云杉立枯病的拮抗作用
-
以沙地云杉立枯病病原真菌为靶标菌,4株沙地云杉内生拮抗菌的发酵液均表现出良好的拮抗活性,菌株SDYS180发酵液的拮抗活性最高,SDYS95的菌株对峙拮抗活性最高,SDYS63的挥发性代谢产物的拮抗活性最高。
4株沙地云杉内生拮抗菌对盆栽沙地云杉立枯病均表现出不同程度的拮抗活性。从病苗形态指标来看,SDYS180对盆栽沙地云杉立枯病的拮抗活性最高;从生理指标来看,拮抗活性最高的是SDYS95。









 DownLoad:
DownLoad: