-
晋西黄土区面临严重的水土流失与土壤退化问题,合理选择植被恢复措施可有效保持水土并改善生态环境,其中营造人工林是该地区生态重建的重要途径[1]。在生态修复过程中,植物与土壤通过动态互作逐步形成不同生态功能特征的土壤生态系统[2]。植物生长依赖土壤养分供给,而不同林分类型的化学组分、微生物群落结构及其代谢活动的差异性,驱动土壤养分状况呈现多样化演变趋势[3]。作为植物根系、土壤动物及微生物活动分泌的高效催化蛋白,土壤胞外酶几乎参与所有土壤生化反应,可提升养分有效性,促进植物吸收利用[4]。曹雨婷等[5]在对比不同菌根类型林分发现:混交林土壤中的β-葡糖苷酶、β-N-乙酰氨基葡萄糖苷酶及亮氨酸氨基肽酶活性显著高于纯林。此外,杨君珑等[6]研究显示:六盘山针阔混交林地的磷酸酶与脲酶活性居7种水源涵养林之首。这种酶活性差异源于不同林分引起的微生物量、种群结构及代谢过程变化[7]。健康土壤微生物群落对土壤结构改良具有双重效应,既优化物理性质,又增强污染物降解能力。反之,土壤微生物多样性的减少可能会对土壤的长期可持续利用产生不利影响[8]。张星星等[9]对亚热带树种的研究表明:林分类型通过调控凋落物数量与质量影响养分供给,进而改变微生物底物有效性并调节酶活性。此外,表层土壤因微生物活动旺盛,其养分含量显著高于深层土壤[10]。不同林分类型通过调控土壤养分、酶活性及微生物群落的协同作用,最终形成差异化的土壤生态功能。
黄土高原面临严峻的环境挑战,如严重土壤侵蚀、低保水性和土壤养分匮乏等问题。针对当地实际情况,迫切需要有效的植被恢复措施来管理。有效营造混交林或纯林一直是该地区生态学研究的热点。以往研究集中比较混交林和纯林在生长和土壤条件上的差异,聚焦于微生物对土壤碳氮动态变化的作用[11]。然而,土壤微生物不仅参与养分循环和有机质的转换,还会通过各种生物化学和物理机制改变土壤生存环境[12]。鉴于此,本研究通过纯林、混交林的土壤养分、酶活性、微生物多样性和组成,主要解决2个关键问题:①混交林是否能够提升土壤肥力和活性,从而缓解黄土区碱性土壤面临的养分匮乏等问题?②林分类型转变如何影响土壤微生物多样性及组成,并与土壤环境因子变化产生关联?本研究旨在为晋西黄土区通过人工造林进行生态恢复提供理论依据。
-
研究区位于山西省吉县蔡家川流域(36°01′13″~36°19′19″N,110°41′17″~110°47′52″E),该区属于黄土高原残塬沟壑区,总面积为39.33 km2,呈东西走向的狭长地形(长度约14 km),海拔高程为900.0~1 513.0 m。区域气候为温带大陆性季风气候,年均气温为10.0 ℃,年均降水为575.9 mm,其中76%的降水集中于6—9月,年蒸发量为1 729.0 mm。成土母质为黄土,发育典型褐土。现存植被以水土保持林为主,包括刺槐Robinia pseudoacacia、油松Pinus tabulaeformis、侧柏Platycladus orientalis等人工纯林及混交林,以及辽东栎Quercus wutaishanica、山杨Populus davidiana等。林下灌木层以黄刺玫Rosa xanthina、杠柳Periploca sepium为优势种,草本层由薹草Carex spp.、茜草Rubia cordifolia等构成。
-
在对研究区进行全面踏查的基础上,选取具有代表性的刺槐纯林、油松纯林、侧柏纯林、油松-刺槐混交林、辽东栎-山杨混交林5种林分类型,以荒草地作为对照。不同林分类型样地选取尽可能保证立地条件(海拔、密度、坡向)和林分特征(林龄、郁闭度、密度等)基本一致,各林分样地基本信息如表1所示。在每个林分中设3个20 m×20 m的大样方,每个样方中随机选取3处具有代表性的取样点,用土钻取0~20 cm表层土壤,将3个取样点的土壤样品混合均匀作为1个样品,去掉土壤样品中的石砾与动植物残体,分2份装入自封袋带回实验室,一份自然风干用于土壤养分、酶活性的测定,另一份立即过2 mm筛用于土壤微生物多样性的测定,设置3个重复。
林分类型 海拔/
m密度/
(株·hm−2)坡度/
(°)坡向 郁闭度/
%林龄/
a胸径/
cm树高/
m土壤含水
量/(g·kg−1)凋落物蓄
积量/(t·hm−2)CH 1 500.9 1 300 20 阳坡 53.4 34 17.08±4.59 9.24±1.13 5.62±1.62 14.48±1.24 YS 1 121.3 1 150 21 阳坡 59.2 33 14.19±2.66 8.48±0.85 6.53±0.25 15.66±2.29 CB 1 123.2 1 325 15 阳坡 41.0 34 10.68±2.75 5.05±0.71 7.85±1.04 13.26±2.28 MF 1 085.7 1 550 25 阳坡 66.0 34 12.76±3.94 9.49±1.27 9.84±0.59 17.95±0.92 CSL 1 047.9 1 400 15 阳坡 63.2 34 10.97±3.46 9.56±1.35 12.07±2.30 32.05±5.01 HC 1 077.0 0 3.74±0.31 说明:CH、YS、CB、MF、CSL、HC分别表示刺槐、油松、侧柏、油松-刺槐混交林、辽东栎-山杨混交林、荒草地。 Table 1. Basic information of forest plots
-
参照《土壤农业化学分析方法》[13],测定土壤全氮(TN)、土壤全磷(TP)、土壤硝态氮(NNC)、土壤铵态氮(ANC)、土壤速效磷(AP)、土壤有机质(SOM)、土壤pH。
-
采用荧光酶标法[14],测定4种土壤酶活性,包括β-葡萄糖苷酶(BG)、β-N-乙酰氨基葡萄糖苷酶(NAG)、亮氨酸氨基肽酶(LAP)、碱性磷酸酶(ALP)。
-
根据 E.Z.N.A.® soil DNA kit说明书进行微生物群落总基因组DNA抽提,使用10 g·L−1的琼脂糖凝胶电泳检测抽提的基因组DNA质量,使用NanoDrop2000测定DNA浓度和纯度。以上述提取的DNA为模板,使用携带Barcode序列的上游引物338F (5′-ACTCCTACGGGAGGCAGCAG-3′)和下游引物806R (5′-GGACTACHVGGGTWTCTAAT-3′)[15]对16S rRNA基因V3~V4可变区进行PCR扩增。利用Illumina Nextseq 2000平台进行测序,并使用USEARCH软件借助DADA2方法进行拼接和过滤,按照≥99%相似度聚类为特征单元(ASV),将其比对至UNITE真菌数据库和SILVA细菌数据库进行物种注释,最后,按最小样本序列将得到的数据抽平后,进行土壤微生物多样性分析,具体分析微生物α多样性和微生物群落组成,测序工作委托上海美吉生物医药科技有限公司完成。
-
采用Excel 2019和SPSS 21.0软件进行数据分析,用Origin 2022绘图。采用单因素方差分析(one-way ANOVA法)评价不同林分类型间土壤养分、酶活性以及微生物多样性的差异,用Duncan法进行多重比较,显著性水平为0.05。采用Pearson相关分析研究土壤微生物多样性与土壤养分、酶活性的相关性,采用RDA冗余分析研究微生物群落组成与土壤环境因子间的相互关系。使用R语言“vegan”包中的“varpart()”函数进行VPA分析,量化不同环境因子(如土壤养分、土壤酶活性等)对微生物群落变异的单独解释度与共同解释度,并计算不同环境因子对微生物群落变异的独立与交互贡献比例。
-
由表2表明:土壤全氮(0.92 g·kg−1)、土壤全磷(0.48 g·kg−1)、土壤硝态氮(9.23 mg·kg−1)、土壤速效磷(1.25 mg·kg−1)、土壤有机质(32.05 g·kg−1)质量分数在辽东栎-山杨混交林中最高。不同林分类型间土壤养分指标所呈现的规律一致,从大到小依次为辽东栎-山杨混交林、油松-刺槐混交林、刺槐纯林、油松纯林、侧柏纯林、荒草地。整体上,林地土壤硝态氮、土壤速效磷、土壤有机质质量分数是荒草地的1.5倍以上。
林分类型 全氮/
(g·kg−1)全磷/
(g·kg−1)硝态氮/
(mg·kg−1)铵态氮/
(mg·kg−1)速效磷/
(mg·kg−1)pH 有机质/
(g·kg−1)CH 0.48±0.06 b 0.43±0.04 a 8.21±2.18 ab 9.27±1.31 bc 0.93±0.12 ab 8.58±0.02 a 18.61±5.89 b YS 0.33±0.22 bc 0.38±0.23 a 7.89±2.43 ab 9.77±1.06 abc 0.86±0.39 ab 8.57±0.02 a 13.90±6.28 bc CB 0.37±0.09 c 0.42±0.09 a 4.59±0.44 bc 8.08±1.74 c 0.56±0.15 b 8.66±0.07 a 8.24±1.63 c MF 0.82±0.05 a 0.43±0.14 a 8.85±2.65 a 12.71±2.02 a 0.99±0.38 ab 8.63±0.01 a 30.04±2.52 a CSL 0.92±0.08 a 0.48±0.13 a 9.23±2.10 a 11.33±2.08 ab 1.25±0.36 a 8.55±0.13 a 32.05±9.93 a HC 0.45±0.06 c 0.35±0.01 a 3.77±1.26 c 7.77±1.00 c 0.56±0.19 b 8.54±0.02 a 8.15±1.16 c 说明:CH、YS、CB、MF、CSL、HC分别表示刺槐、油松、侧柏、油松-刺槐混交林、辽东栎-山杨混交林、荒草地。不同小写字母表示不同林分类型间差异显著(P<0.05)。 Table 2. Soil nutrient content in different forest stand types
-
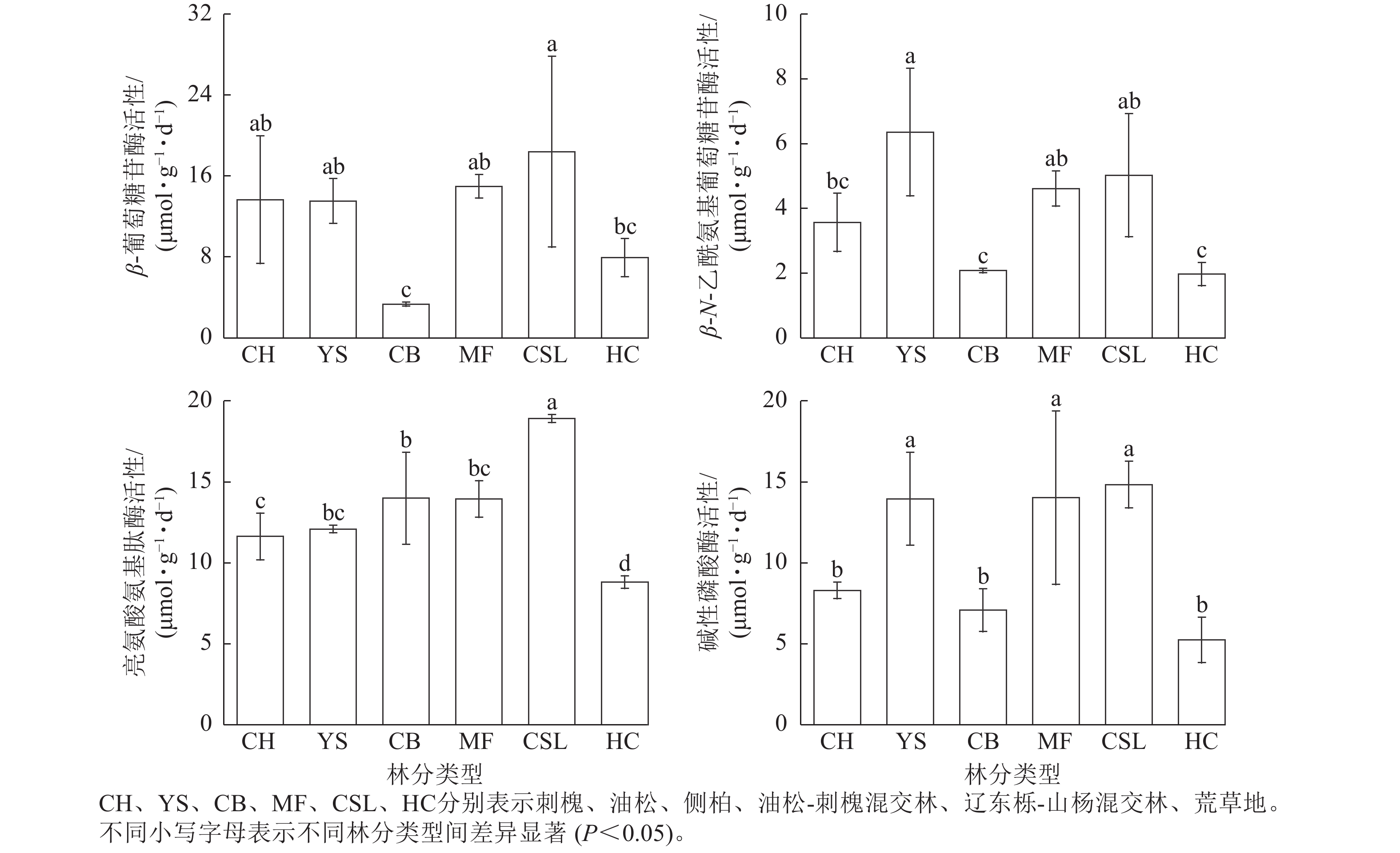
由图1表明:土壤β-葡萄糖苷酶、亮氨酸氨基肽酶、碱性磷酸酶活性在辽东栎-山杨天然混交林中最高,分别为18.41、18.92、14.84 μmol·g−1·d−1,分别为荒草地的2.23、2.15、2.83倍。土壤β-N-乙酰氨基葡萄糖苷酶活性在油松纯林中最高,为6.36 μmol·g−1·d−1,是荒草地的3.23倍。侧柏纯林的土壤β-葡萄糖苷酶活性低于荒草地,仅为荒草地的40%。除了油松纯林的土壤β-N-乙酰氨基葡萄糖苷酶活性和荒草地的土壤β-葡萄糖苷酶活性外,土壤β-葡萄糖苷酶、β-N-乙酰氨基葡萄糖苷酶、亮氨酸氨基肽酶、碱性磷酸酶活性在不同林分类型间表现出相似的规律性,从大到小依次为混交林、纯林、荒草地。
-
由表3表明:5种不同林分类型土壤真菌的ACE和Chao1指数从大到小依次为油松-刺槐混交林、辽东栎-山杨混交林、侧柏纯林、刺槐纯林、油松纯林,其中辽东栎-山杨混交林的ACE和Chao1指数均显著高于其他林分(P<0.05)。油松纯林的Simpson指数高于其他林分,是荒草地的5.4倍,但Shannon指数最低,不到混交林的70%。
林分类型 ACE指数 Chao1指数 Shannon指数 Simpson指数 CH 398.81±88.37 b 398.59±90.38 b 3.80±0.65 c 0.064 9±0.050 0 ab YS 251.91±49.91 c 251.27±49.87 c 3.02±0.31 d 0.113 1±0.044 6 a CB 468.18±49.78 b 468.02±49.79 b 4.65±0.16 ab 0.030 8±0.006 2 b MF 677.11±51.03 a 676.53±50.74 a 5.02±0.16 a 0.014 0±0.003 1 b CSL 505.53±113.37 b 505.10±113.07 b 4.15±0.39 bc 0.047 5±0.025 2 b HC 471.17±56.28 b 470.79±56.30 b 4.71±0.31 ab 0.021 0±0.008 6 b 说明:CH、YS、CB、MF、CSL、HC分别表示刺槐、油松、侧柏、油松-刺槐混交林、辽东栎-山杨混交林、荒草地。不同小写字母表示不同林分类型间差异显著(P<0.05)。 Table 3. Soil fungal diversity in different forest stand types
由表4表明:5种不同林分类型土壤细菌的ACE和Chao1指数从大到小依次为侧柏纯林、刺槐纯林、油松-刺槐混交林、辽东栎-山杨混交林、油松纯林,其中侧柏纯林的ACE和Chao1指数显著高于油松纯林(P<0.05)。油松纯林的Simpson指数最高,显著高于侧柏纯林、辽东栎×山杨混交林和荒草地(P<0.05),是它们的1.3倍,Shannon指数最低。除Simpson指数外,土壤细菌的其他3种指数均高于土壤真菌,ACE和Chao1指数更是高出2倍以上。
林分类型 ACE指数 Chao1指数 Shannon指数 Simpson指数 CH 1 705.36±233.99 ab 1 697.90±236.41 ab 6.83±0.13 ab 0.001 8±0.000 2 ab YS 1 361.93±52.44 b 1 355.59±53.41 b 6.62±0.07 b 0.002 1±0.000 1 a CB 1 844.42±383.68 a 1 840.13±389.50 a 6.91±0.22 a 0.001 6±0.000 3 b MF 1 525.60±154.26 ab 1 518.86±144.67 ab 6.74±0.04 ab 0.001 9±0.000 1 ab CSL 1 483.60±105.52 ab 1 475.22±102.09 ab 6.78±0.07 ab 0.001 6±0.000 1 b HC 1 600.95±14.91 ab 1 596.63±15.25 ab 6.87±0.02 a 0.001 6±0.000 1 b 说明:CH、YS、CB、MF、CSL、HC分别表示刺槐、油松、侧柏、油松-刺槐混交林、辽东栎-山杨混交林、荒草地。不同小写字母表示不同林分类型间差异显著(P<0.05)。 Table 4. Soil bacterial diversity in different forest stand types
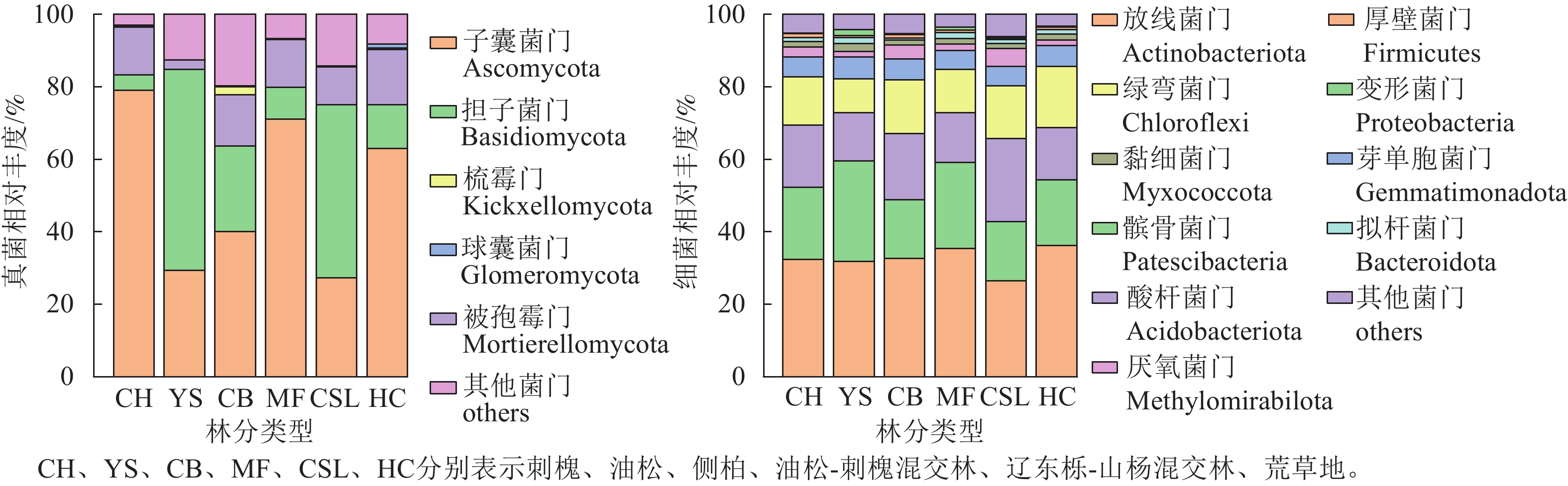
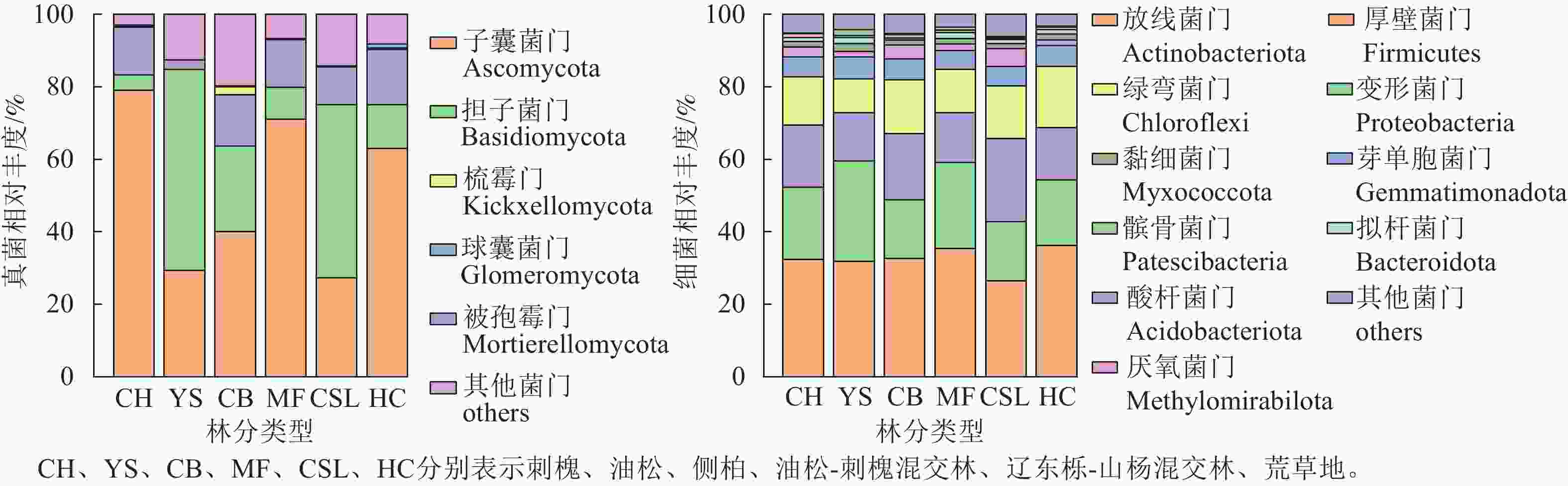
研究区不同林分类型土壤微生物群落组成分析(图2)显示:真菌优势菌群为子囊菌门Ascomycota (27.3%~78.99%)和担子菌门Basidiomycota (4.24%~55.56%)。相较于荒草地,子囊菌门相对丰度在刺槐纯林和油松-刺槐混交林中有所提高,而油松纯林、侧柏纯林和辽东栎-山杨混交林分别降低53.55%、36.47%和56.69%;担子菌门相对丰度在油松、侧柏和辽东栎-山杨混交林中较荒草地分别增长3.61、0.95和2.96倍,而刺槐和油松-刺槐混交林则减少。相较于荒草地,被孢霉门Mortierellomycota在所有林分中均呈下降趋势(降幅为2.56%~15.19%)。细菌优势菌群为放线菌门Actinobacteriota (26.4%~36.19%)、变形菌门Proteobacteria (16.21%~27.73%)、酸杆菌门Acidobacteriota (13.34%~22.87%)和绿弯菌门Chloroflexi (9.42%~16.86%)。细菌群落的相对丰度在各林分间均差异不明显。
-
相关分析(表5)表明:土壤β-葡萄糖苷酶与土壤全氮呈显著正相关(P<0.05),与土壤铵态氮、土壤硝态氮、土壤速效磷、土壤有机质呈极显著正相关(P<0.01)。土壤β-N-乙酰氨基葡萄糖苷酶与土壤速效磷呈显著正相关(P<0.05),与土壤硝态氮呈极显著正相关(P<0.01)。土壤亮氨酸氨基肽酶与土壤铵态氮、土壤速效磷呈显著正相关(P<0.05),与土壤全氮、土壤有机质呈极显著正相关(P<0.01)。土壤碱性磷酸酶与土壤全氮、土壤铵态氮、土壤硝态氮呈显著正相关(P<0.05),与土壤速效磷、土壤有机质呈极显著正相关(P<0.01)。土壤真菌ACE、Chao l指数均与土壤全氮呈显著正相关(P<0.05)。细菌Simpson指数与土壤养分无显著相关性,ACE、Chao l、Shannon指数与土壤硝态氮呈显著负相关(P<0.05)。
指标 全氮 全磷 铵态氮 硝态氮 速效磷 pH 有机质 土壤酶 β-葡萄糖苷酶 0.54* 0.19 0.60** 0.75** 0.64** −0.22 0.63** β-N-乙酰氨基葡萄糖苷酶 0.24 0.06 0.46 0.72** 0.58* −0.13 0.44 亮氨酸氨基肽酶 0.60** 0.37 0.55* 0.37 0.51* 0.18 0.65** 碱性磷酸酶 0.53* 0.06 0.58* 0.57* 0.74** 0.02 0.62** 真菌 ACE 0.57* −0.05 0.37 0.01 0.13 0.10 0.38 Chao1 0.57* −0.05 0.37 0.01 0.13 0.10 0.38 Shannon 0.31 −0.04 0.06 −0.26 −0.22 0.19 0.03 Simpson −0.27 0.01 −0.01 0.23 0.27 −0.11 −0.02 细菌 ACE −0.16 −0.13 −0.31 −0.51* −0.19 0.27 −0.25 Chao1 −0.16 −0.13 −0.31 −0.51* −0.20 0.27 −0.25 Shannon −0.02 −0.06 −0.37 −0.49* −0.20 0.12 −0.20 Simpson −0.25 −0.01 0.22 0.37 −0.04 0.01 −0.02 说明:*表示显著相关 (P<0.05);**表示极显著相关 (P<0.01)。 Table 5. Correlation between soil nutrients, soil enzyme activity and microbial diversity
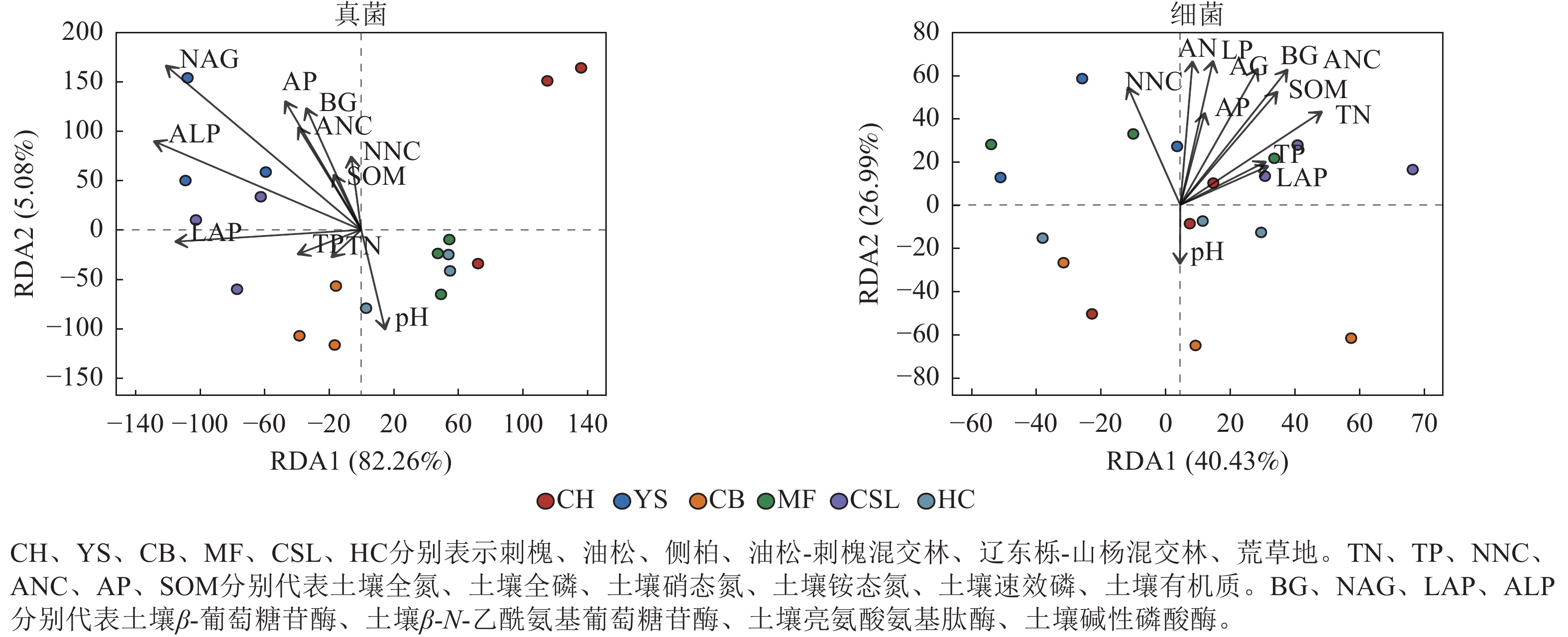
冗余分析(图3)显示:前2个轴分别解释了受土壤环境因子影响的真菌群落组成和细菌组成的方差,分别为87.34%和67.42%。土壤真菌群落与土壤β-N-乙酰氨基葡萄糖苷酶、土壤碱性磷酸酶呈显著相关(P<0.05),土壤细菌群落与土壤全氮、土壤铵态氮、土壤有机质、土壤β-葡萄糖苷酶、土壤β-N-乙酰氨基葡萄糖苷酶、土壤碱性磷酸酶呈显著相关(P<0.05)。土壤β-N-乙酰氨基葡萄糖苷酶、土壤铵态氮分别是真菌、细菌群落解释率最高的环境因子(表6),表明这2个因子对真菌、细菌群落的分布和多样性有显著影响。
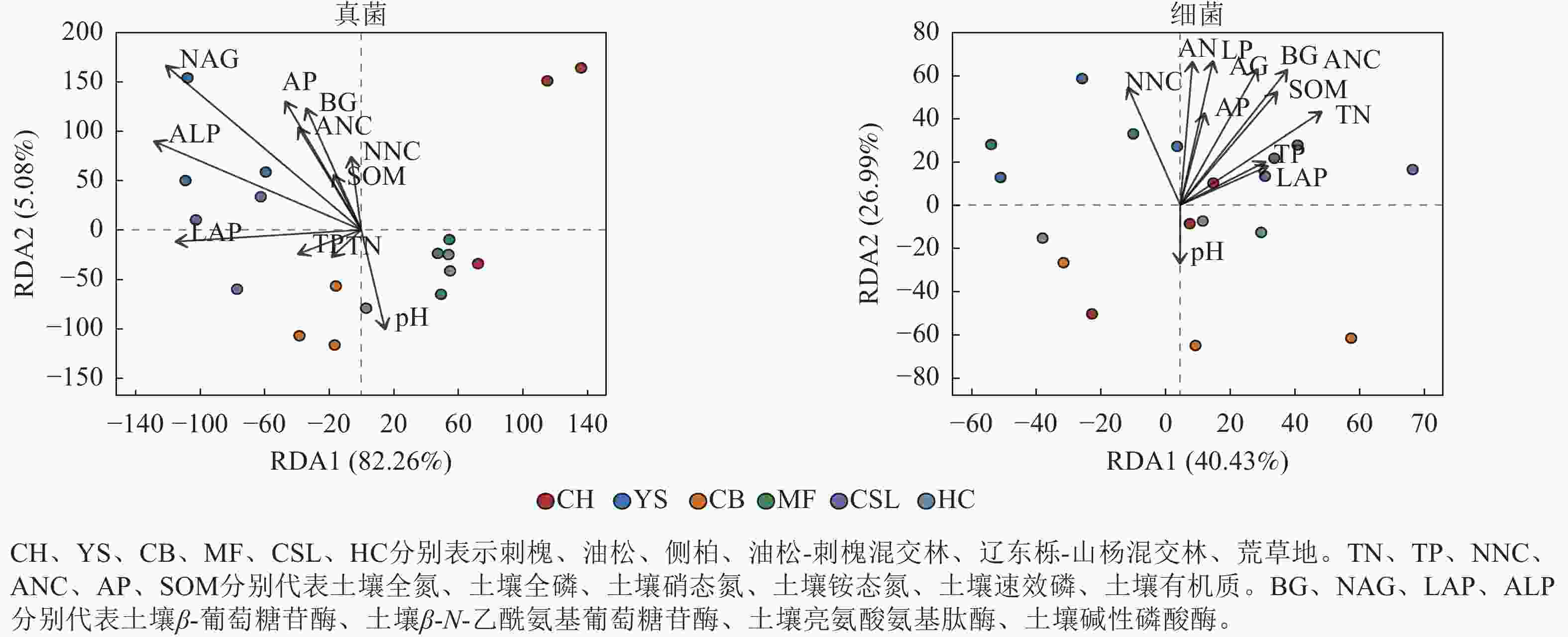
Figure 3. Redundancy analysis (RDA) of soil microbial community composition with environmental factors
群落 指标 RDA1 RDA2 解释率 P 群落 指标 RDA1 RDA2 解释率 P 真菌 TN −0.62 −0.79 0.01 0.93 细菌 TN 0.69 0.73 0.39 0.02* TP −0.92 −0.39 0.02 0.84 TP 0.76 0.65 0.11 0.39 NNC −0.21 0.98 0.06 0.62 NNC −0.17 0.99 0.30 0.07 ANC −0.49 0.87 0.15 0.29 ANC 0.49 0.87 0.53 0.00** AP −0.48 0.88 0.23 0.12 AP 0.26 0.97 0.19 0.21 SOM −0.44 0.90 0.04 0.74 SOM 0.52 0.86 0.39 0.02* pH 0.28 −0.96 0.12 0.39 pH −0.11 −0.99 0.07 0.61 BG −0.41 0.91 0.19 0.20 BG 0.40 0.91 0.48 0.00** NAG −0.69 0.72 0.57 0.00** NAG 0.24 0.97 0.47 0.01** LAP −1.00 0.01 0.18 0.23 LAP 0.79 0.61 0.10 0.45 ALP −0.86 0.51 0.35 0.03* ALP 0.16 0.99 0.45 0.01* 说明:TN、TP、NNC、ANC、AP、SOM分别代表土壤全氮、土壤全磷、土壤硝态氮、土壤铵态氮、土壤速效磷、土壤有机质。BG、NAG、LAP、ALP分别代表土壤β-葡萄糖苷酶、土壤β-N-乙酰氨基葡萄糖苷酶、土壤亮氨酸氨基肽酶、土壤碱性磷酸酶。*表示解释显著(P<0.05);**表示解释极显著(P<0.01)。 Table 6. Explanatory rate of soil microbial communities by environmental factors
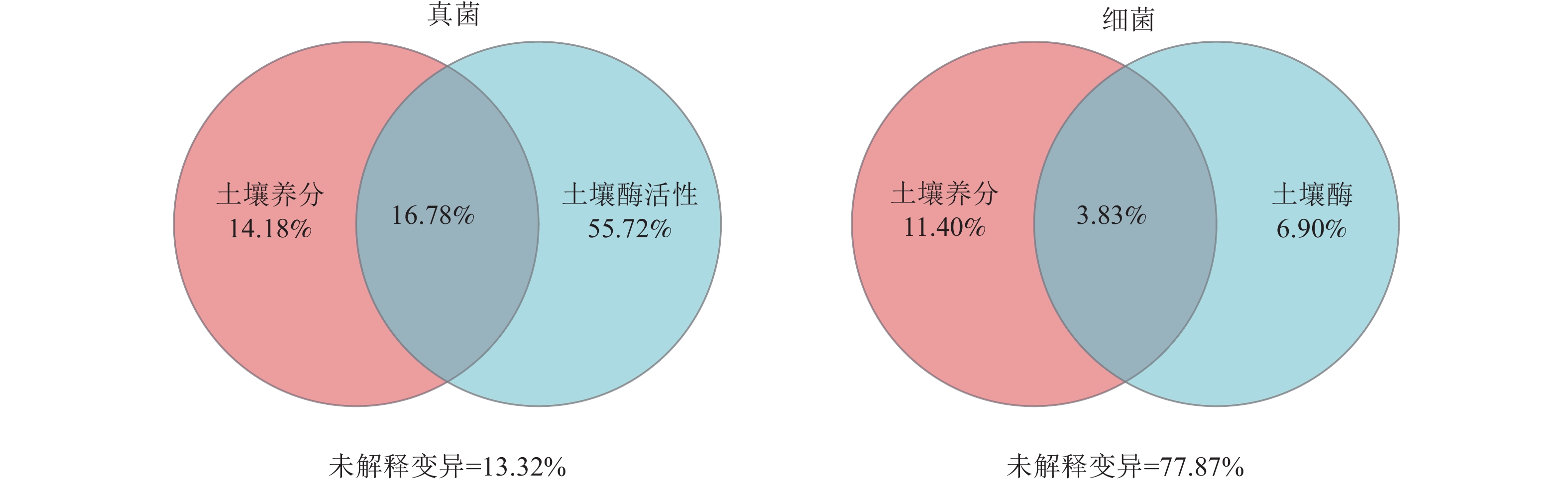
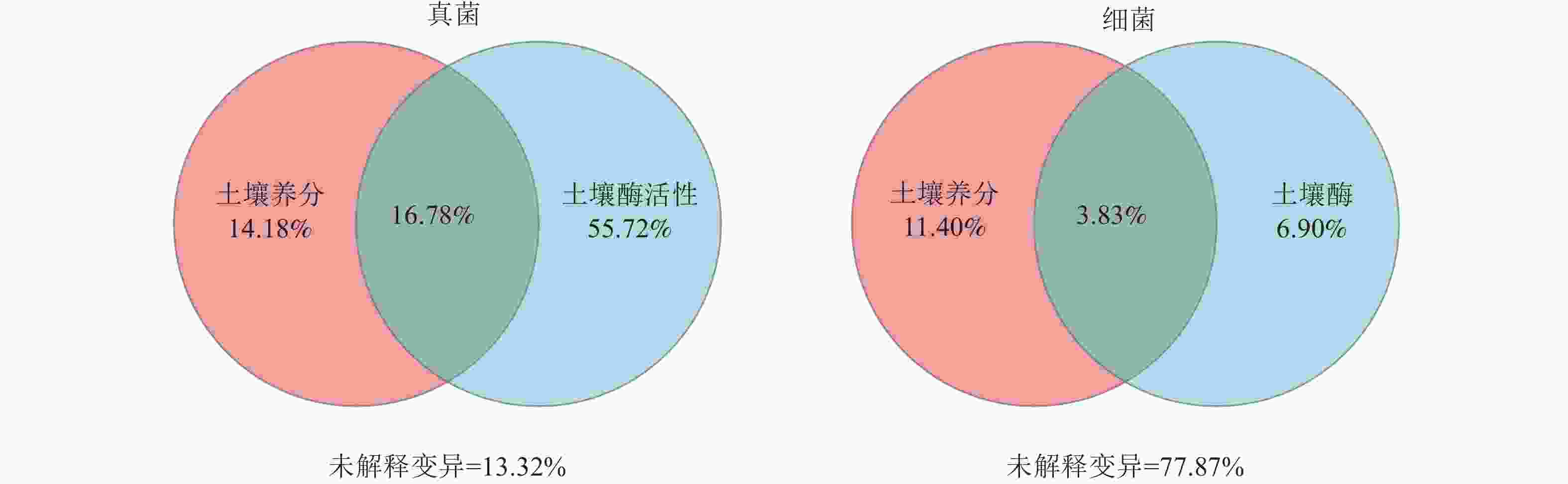
方差分解分析(图4)显示:在真菌群落中,土壤酶活性的解释度为72.50%,高于土壤养分含量的解释度(30.96%)。在细菌群落中,土壤酶活性的解释度为10.73%,低于土壤养分含量的解释度(15.23%)。此外,土壤养分含量和酶活性对真菌群落的共同解释度为16.78%,高于对细菌群落的共同解释度(3.83%),表明土壤养分含量和酶活性在真菌群落中具有较强的协同作用。
-
本研究表明:油松-刺槐混交林和辽东栎-山杨混交林中土壤全氮和土壤有机质质量分数显著高于纯林及荒草地,这可能与2种混交林中较高的凋落物蓄积量有关,这些凋落物在微生物的作用下逐步分解,促进了养分循环,从而提高了土壤养分水平[16]。相比之下,荒草地的养分含量最低,但全氮质量分数高于油松纯林和侧柏纯林。这可能是由于荒草地生态系统中,浅根系草本植物与拟杆菌门Bacteroidaceae形成的根际共生固氮体系虽可促进表层氮素短期积累,但冠层结构的缺失显著加剧了降雨驱动的养分淋失过程,导致荒地的养分含量较低[17−18]。此外,土壤全磷质量分数在各林分类型间无显著差异,这可能是全磷更易受水分和气温等环境因素的影响,对林分类型变化的响应较小[19]。
土壤胞外酶作为促进有机质分解的催化剂,在生态系统物质循环和能量流动中扮演着重要的角色[20]。本研究发现:不同林分类型对土壤酶活性有显著影响,辽东栎-山杨混交林的土壤β-葡萄糖苷酶、土壤亮氨酸氨基肽酶和土壤碱性磷酸酶活性显著高于其他林分,这可能是因为辽东栎-山杨混交林土壤具有较高的养分水平和水分条件,可为微生物提供良好的生长环境,从而促进土壤胞外酶的产生[21]。与此相比,油松纯林的土壤β-N-乙酰氨基葡萄糖苷酶活性最高,这可能是因为其分泌的凋落物含有的木质素、纤维素等物质比例更适合β-N-乙酰氨基葡萄糖苷酶参与分解,为土壤微生物提供了丰富的碳源,刺激微生物分泌更多的β-N-乙酰氨基葡萄糖苷酶来分解这些物质,这与马园园等[22]的研究结果相一致。
-
林分类型的变化对土壤微生物α多样性具有显著影响。本研究发现:在真菌群落中,油松-刺槐混交林的ACE、Chao1和Shannon指数显著增加,这与以往的研究一致,可能是因为混交林中具有更丰富的营养资源和复杂的栖息环境,促进了土壤真菌丰富度和多样性的提升[23]。在细菌群落中,侧柏纯林的ACE、Chao1和Shannon指数显著高于其他林分,与前人研究结果存在一定的差异[24],这可能是因为侧柏凋落物的分解产生适合特定细菌群落的碳源和氮源,形成稳定的微生物环境,从而支持更多专一性细菌的生长[25]。
进一步分析表明:林分类型的变化对土壤微生物群落组成具有显著影响,尤其是对真菌类群的相对丰度。在真菌群落中,子囊菌门、担子菌门为优势类群。本研究发现刺槐纯林和油松-刺槐混交林中,子囊菌门相对丰度显著提高,而在油松纯林、侧柏纯林和辽东栎-山杨混交林中则有所下降,这可能是因为子囊菌门能够分泌降解易分解有机质的酶类,而刺槐作为固氮树种,其凋落物富含氮元素且易于分解,这为子囊菌门的生长提供了有利的营养条件[26]。而担子菌门相对丰度的结果与之相反,这可能是因为担子菌门可以降解较难分解的木质纤维素,而针叶树种(侧柏、油松)的凋落物中富含蜡质和难降解的木质素,从而促进了担子菌门的生长[27]。细菌群落中,变形菌门、酸杆菌门、放线菌门和绿弯菌门为优势菌门,其相对丰度在各林分间无明显差异。这表明细菌群落对林分类型的变化具有较强的适应性,这主要得益于细菌在分解复杂有机物和维持养分循环方面的生态功能特性,使其能够在不同的环境条件下生存并持续发挥作用[28]。
-
有研究表明:土壤养分与酶活性存在显著或极显著的相关性[29],这与本研究结果相一致。此外,本研究还发现:真菌群落与土壤β-N-乙酰氨基葡萄糖苷酶、土壤碱性磷酸酶呈显著相关,但与土壤养分含量并无明显相关性,而细菌群落与土壤全氮、土壤铵态氮、土壤有机质、土壤β-葡萄糖苷酶、土壤β-N-乙酰氨基葡萄糖苷酶、土壤碱性磷酸酶呈显著相关,表明真菌和细菌群落在土壤养分需求和响应上存在差异[30]。有研究指出细菌群落对氮和磷的响应比真菌群落更为敏感[31],本研究结果进一步验证了这一观点。
方差分解分析表明:土壤酶活性对真菌群落变化的解释度显著高于对细菌群落的解释度,这表明在黄土高原典型林地土壤中,真菌群落的结构更容易受到土壤酶活性的驱动[32]。此外,土壤养分和酶活性对真菌群落的联合解释度明显高于对细菌群落的联合解释度,该结果进一步表明真菌群落受到土壤养分含量和酶活性的共同调控[10]。
-
与纯林相比,混交林显著提高了土壤养分含量和酶活性,表明单一树种的种植已难以有效解决晋西黄土区面临的土壤养分匮乏等问题,而多样化的树种组成更有助于改善土地肥力,增强林地生态系统的稳定性。林分类型显著影响微生物多样性,尤其是在混交林中,真菌多样性得到显著增加,这有助于增强复杂有机物的分解能力,促进土壤养分循环,优化土壤生态功能。建议在晋西黄土区植被恢复和重建中,优先考虑落叶阔叶或针叶阔叶树种为核心的混交林配置方案,这种混交模式在提升土壤肥力的同时,可优化微生物群落结构,从而增强林地生态系统的稳定性与可持续发展能力。
Differential analysis of soil nutrient-enzyme activity-microbial diversity in typical woodland in loess area
doi: 10.11833/j.issn.2095-0756.20250224
- Received Date: 2025-03-25
- Accepted Date: 2025-11-07
- Rev Recd Date: 2025-10-30
- Available Online: 2026-04-02
- Publish Date: 2026-04-02
-
Key words:
- forest stand type /
- soil nutrients /
- enzyme activity /
- microbial diversity /
- loess area of Western Shanxi
Abstract:
| Citation: | LI Zhipeng, BI Huaxing, GUAN Ning, et al. Differential analysis of soil nutrient-enzyme activity-microbial diversity in typical woodland in loess area[J]. Journal of Zhejiang A&F University, 2026, 43(2): 370−380 doi: 10.11833/j.issn.2095-0756.20250224 |









 DownLoad:
DownLoad: